BECTON, DICKINSON & COMPANY
872 Marks
Corp.
First Filed:Oct 5, 1917Latest Filed:Mar 19, 2026Address:1 Becton Drive, Franklin Lakes, NJ 07417-1880, US
Portfolio Overview
Previous
155Top Classes
Class 10
Medical ApparatusClass 9
Electronics, Software & Scientific EquipmentClass 1
Industrial Chemicals & AdhesivesTTAB Proceedings
Total Proceedings
68As Plaintiff
42(62%)As Defendant
26(38%)Top Firms
Loading...
Trademark Activity Timeline(1980 – 2026)
Trademark Portfolio
1,027 results
(current & previous marks)
Refine by Mark/Description🔽
Filed
Registered
Last Activity
Filters
Ownership
872
155
Status
237
26
371
249
144
Mark Type
748
42
21
216
0
136
5
2
108
32

Goods & Services
Class 005: Solutions for cleansing the skin for medical use; Products and Solutions for cleansing the skin for medical use; Chemical Solutions for pharmaceutical or medical purposes, namely, for cleansing for OBGYN procedures

Goods & Services
Class 009: Downloadable and recorded computer software for use in medication management, drug diversion investigations, pharmacy operations and analytics, and healthcare workflow optimization; Downloadable and recorded software for clinical decision support, infection prevention analytics, antimicrobial and antibiotic stewardship, and patient safety analytics.; Downloadable and recorded software for monitoring, detecting, and analyzing potential controlled substance diversion and identifying patients at risk for adverse drug events.; Downloadable and recorded software for inventory management, inventory forecasting, inventory optimization, and supply chain analytics in healthcare facilities.; Downloadable computer dashboards and data visualization software for aggregating and displaying healthcare operational data.; Downloadable software for integrating medical devices, medication management systems, and electronic medical records.; Class 035: Business consulting services in the field of healthcare inventory management, pharmacy operations, and workflow optimization.; Business risk management consulting relating to controlled substance diversion detection and investigation workflows.; Business data analysis services in the field of healthcare operations, medication usage, and inventory performance.; Class 042: Software as a service (SaaS) featuring software for medication management, drug diversion investigations, pharmacy operations and analytics, clinical decision support, infection prevention analytics, and healthcare workflow optimization.; SaaS featuring software for detecting, monitoring, and analyzing potential controlled substance diversion and identifying patients at risk for adverse drug events.; SaaS featuring software for healthcare inventory management, inventory optimization, and supply chain analytics.; Providing temporary use of non downloadable software for aggregating, analyzing, and visualizing healthcare operational data.; Electronic data integration services for connecting medical devices, medication management systems, and electronic medical records.; Providing online non downloadable computer dashboards for viewing and managing healthcare operational metrics.

Goods & Services
Class 010: Medical injectors, sold empty, for use in connection with sterile disposable systems, namely, injectors for attaching to drug vials, syringes or infusion tubing to seal off hazardous drugs or vapor concentrations; closed system drug-transfer devices, namely, medical injectors, sold empty, for connecting to drug vials, syringes, or infusion tubing to prevent transfer of environmental contaminants; medical injector and connector devices, sold empty, for the sealed handling of parenteral drugs

Goods & Services
Class 010: Medical devices and apparatus, namely, intravenous catheters, and parts and fittings therefor, excluding reperfusion catheters and associated connectors, fittings and adaptors for reperfusion catheters; Needleless access devices and systems, namely, valves, adapters, and extension sets for drawing blood; needleless drug delivery devices sold empty in the nature of catheters, cannulas, tubing, connectors, valves, adapters, and extension sets; needleless injection systems in the nature of injectors, catheters, cannulas, tubing, connectors, valves, adapters, and extension sets; needleless injectors for medical use; needleless syringes; needleless connectors, valves, adapters, and extension sets for drawing blood; needleless intravenous access products, namely, cannulas, injection site syringes, drug vial adapters, catheters, and intravenous tubing; needles for medical use; needles and parts and fittings therefor for medical use; catheters and parts and fittings therefor; all the aforementioned goods for medical, dental, or veterinary use; Medical devices and apparatus, comprised of an ultrasound machine, ultrasound probes, ultrasound probe covers, recorded medical imaging software, an activator for magnetizing the needle, and also including electrical power supply, roll stands especially adapted for mounting medical devices and replacement parts and fittings for the ultrasound machine; Class 044: Providing medical information, education and training

Goods & Services
Class 009: Downloadable software for processing, managing, retrieving and storing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument operational data, and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; downloadable software for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; downloadable software for use in the life sciences industry to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems; downloadable software for analyzing health, medical, and pharmaceutical data and medical device data and medical diagnostic instrument data; electronic control devices, apparatus and computer hardware for storing and dispensing pharmaceuticals, and parts and fittings therefor; Class 010: Medical devices, namely a full line of medical devices and instruments for use in surgery and surgical apparatus and instruments for medical use; medical devices, namely infusion and injection devices for administering drugs, infusion pumps and controllers, namely, volumetric infusion pumps, syringe pumps, programmable infusion pumps, programmable syringe pumps and the resident control programs; clinical vital signs measurement instruments, namely, thermometers for medical use, disposable thermometer covers, blood pressure measurement instruments, pulse rate measurement instruments, blood pressure cuffs, pulse oximetry instruments, namely, sensors and monitors, respiration measurement instruments, and ecg instruments in the nature of electrocardiographic recorders; medical fluid administration sets for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves; medical devices for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves; intravenous fluid containers sold empty, monitors, alarms, rate meters; sphygmomanometers; enteric infusion pumps and bags, sold empty, therefor; medical instrument and intravenous fluid container stands and hanger devices in the nature of intravenous stands and poles and stands and hangers specifically adapted for patient infusion systems consisting primarily of infusion pumps and controllers; patient monitors for critical care; medical apparatus and instruments for patient monitoring; medical apparatus and instruments for monitoring patient hemodynamics and oxygen saturation; medical diagnostic equipment, namely, medical instruments for the detection of infectious diseases, and parts and fittings therefor; medical diagnostic equipment, namely, medical instruments for the analysis of body fluid, and parts and fittings therefor; Class 042: Platform as a service (PAAS) featuring computer software platforms for processing, managing, retrieving and storing health, medical and medical device and medical diagnostic instrument operational data and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; Platform as a service (PAAS) featuring computer software platforms for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; Platform as a service (PAAS) featuring computer software platforms for use to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; software as a service (SAAS) featuring software for analyzing medical and medical device and medical diagnostic instrument operational data and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; software as a service (SAAS) featuring software for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; software as a service (SAAS) featuring software for use to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; platform as a service (PAAS) featuring computer software platforms for analysing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument data; software as a service (SAAS) featuring software for analysing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument data

Goods & Services
Class 009: Downloadable software for processing, managing, retrieving and storing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument operational data, and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; downloadable software for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; downloadable software for use in the life sciences industry to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems; downloadable software for analyzing health, medical, and pharmaceutical data and medical device data and medical diagnostic instrument data; electronic control devices, apparatus and computer hardware for storing and dispensing pharmaceuticals, and parts and fittings therefor; Class 010: Medical devices, namely a full line of medical devices and instruments for use in surgery and surgical apparatus and instruments for medical use; medical devices, namely infusion and injection devices for administering drugs, infusion pumps and controllers, namely, volumetric infusion pumps, syringe pumps, programmable infusion pumps, programmable syringe pumps and the resident control programs; clinical vital signs measurement instruments, namely, thermometers for medical use, disposable thermometer covers, blood pressure measurement instruments, pulse rate measurement instruments, blood pressure cuffs, pulse oximetry instruments, namely, sensors and monitors, respiration measurement instruments, and ecg instruments in the nature of electrocardiographic recorders; medical fluid administration sets for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves; medical devices for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves; intravenous fluid containers sold empty, monitors, alarms, rate meters; sphygmomanometers; enteric infusion pumps and bags, sold empty, therefor; medical instrument and intravenous fluid container stands and hanger devices in the nature of intravenous stands and poles and stands and hangers specifically adapted for patient infusion systems consisting primarily of infusion pumps and controllers; patient monitors for critical care; medical apparatus and instruments for patient monitoring; medical apparatus and instruments for monitoring patient hemodynamics and oxygen saturation; medical diagnostic equipment, namely, medical instruments for the detection of infectious diseases, and parts and fittings therefor; medical diagnostic equipment, namely, medical instruments for the analysis of body fluid, and parts and fittings therefor; Class 042: Platform as a service (PAAS) featuring computer software platforms for processing, managing, retrieving and storing health, medical and medical device and medical diagnostic instrument operational data and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; Platform as a service (PAAS) featuring computer software platforms for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; Platform as a service (PAAS) featuring computer software platforms for use to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; software as a service (SAAS) featuring software for analyzing medical and medical device and medical diagnostic instrument operational data and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; software as a service (SAAS) featuring software for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; software as a service (SAAS) featuring software for use to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; platform as a service (PAAS) featuring computer software platforms for analysing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument data; software as a service (SAAS) featuring software for analysing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument data

Goods & Services
Class 009: Downloadable software for processing, managing, retrieving and storing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument operational data, and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; downloadable software for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; downloadable software for use in the life sciences industry to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems; downloadable software for analyzing health, medical, and pharmaceutical data and medical device data and medical diagnostic instrument data; electronic control devices, apparatus and computer hardware for storing and dispensing pharmaceuticals, and parts and fittings therefor; Class 010: Medical devices, namely a full line of medical devices and instruments for use in surgery and surgical apparatus and instruments for medical use; medical devices, namely infusion and injection devices for administering drugs, infusion pumps and controllers, namely, volumetric infusion pumps, syringe pumps, programmable infusion pumps, programmable syringe pumps and the resident control programs; clinical vital signs measurement instruments, namely, thermometers for medical use, disposable thermometer covers, blood pressure measurement instruments, pulse rate measurement instruments, blood pressure cuffs, pulse oximetry instruments, namely, sensors and monitors, respiration measurement instruments, and ecg instruments in the nature of electrocardiographic recorders; medical fluid administration sets for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves; medical devices for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves; intravenous fluid containers sold empty, monitors, alarms, rate meters; sphygmomanometers; enteric infusion pumps and bags, sold empty, therefor; medical instrument and intravenous fluid container stands and hanger devices in the nature of intravenous stands and poles and stands and hangers specifically adapted for patient infusion systems consisting primarily of infusion pumps and controllers; patient monitors for critical care; medical apparatus and instruments for patient monitoring; medical apparatus and instruments for monitoring patient hemodynamics and oxygen saturation; medical diagnostic equipment, namely, medical instruments for the detection of infectious diseases, and parts and fittings therefor; medical diagnostic equipment, namely, medical instruments for the analysis of body fluid, and parts and fittings therefor; Class 042: Platform as a service (PAAS) featuring computer software platforms for processing, managing, retrieving and storing health, medical and medical device and medical diagnostic instrument operational data and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; Platform as a service (PAAS) featuring computer software platforms for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; Platform as a service (PAAS) featuring computer software platforms for use to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; software as a service (SAAS) featuring software for analyzing medical and medical device and medical diagnostic instrument operational data and hosting applications in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; software as a service (SAAS) featuring software for use in collecting information and data regarding pharmaceutical inventory, dispensing transactions, apparatus availability to optimize efficiency in the field of pharmaceuticals; software as a service (SAAS) featuring software for use to enhance the efficiency of clinical diagnostics and laboratory operations and to improve patient outcomes by facilitating data analysis and workflow management across connected instruments and computer systems in the fields of medicine, health, pharmaceuticals, medical devices and medical laboratory diagnostic instruments; platform as a service (PAAS) featuring computer software platforms for analysing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument data; software as a service (SAAS) featuring software for analysing health, medical, and pharmaceutical data and medical device and medical diagnostic instrument data

Goods & Services
Prefillable medical syringes, sold empty

Goods & Services
Charitable foundation services, namely, providing financial assistance for programs and services of others, providing financial assistance to promote health equity initiatives and disaster relief, providing financial assistance to nonprofit global public health organizations; "FOUNDATION"

Goods & Services
Medical devices and instruments, namely, blood collection sets comprising medical needles, luer, wings, tubing, holder and shield, and parts and fittings therefor

Goods & Services
Recorded and downloadable software for capturing and sharing patient infusion data and catheter site data; Medical instruments and equipment, namely, patient infusion systems comprising infusion pumps and controllers, syringe pumps, thermometers for medical use, disposable thermometer covers, blood pressure measurement instruments, pulse rate measurement instruments, blood pressure cuffs, pulse oximetry instruments, namely, sensors and monitors, respiration measurement instruments, and ECG instruments in the nature of electrocardiographic recorders, medical fluid administration sets for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves; medical devices for the delivery of medical fluids, namely, drug delivery tubing, clamps, flow control devices, drug infusion connectors, adapters, injection sites, needleless connectors, needleless ports, needleless injection sites, and medical valves, intravenous fluid containers sold empty, monitors, alarms, rate meters, sphygmomanometers; enteric infusion pumps and bags, sold empty, therefor; medical instrument and intravenous fluid container stands and hanger devices in the nature of intravenous stands and poles and stands and hangers specifically adapted for patient infusion systems consisting primarily of infusion pumps and controllers; gastrointestinal feeding tubes in the nature of tubes for enteral feeding, needle catheter jejunostomy kits comprised of tubing for use with catheters and needles for medical use, specimen holders for medical use in the nature of specimen cup holders, cold and hot packs for chemically producing and absorbing heat for use in medical treatment and therapy; and parts and fittings therefor
Goods & Services
Medical instruments and apparatus, namely, needles and parts and fittings therefor

Goods & Services
Educational services, namely, providing training in the field of infusion therapy; developing printed and digital educational materials for others in the field of infusion therapy; disseminating printed and digital educational materials of others in the field of infusion therapy; conducting lectures, webcasts, and workshops on infusion therapy; "INFUSION"

Goods & Services
Medical and surgical instruments and apparatus, namely, medical and surgical devices for managing and measuring the administration of injectable medications with integrated recorded operating system software

Goods & Services
Artificial material for use in breast surgery, namely, surgical breast implants comprised primarily of artificial material; implantable scaffolds comprising of artificial material for replacing breast tissue or enhancing breast volume; surgical mesh comprised primarily of artificial materials for soft tissue reconstruction; surgical apparatus for breast surgery; and percutaneous breast tissue markers for use in medical diagnosis and treatment
Goods & Services
Medical devices and apparatus, namely, needleless access devices in the nature of needleless vascular access ports for medical use, needleless blood drawing devices, blood collection needles and tubes, and blood drawing devices; phlebotomy devices, namely, blood drawing tubes, needles, winged needle sets comprised of winged blood collection needles and blood collecting tubes, syringes for medical use; needleless injection devices for pharmaceuticals or IV (intravenous) fluids for medical use, needleless adapters in the nature of Luer tapers for medical use, needleless connectors in the nature of Luer tapers for medical use, medical fluid connectors in the nature of Luer tapers for medical use, drug infusion connectors in the nature of Luer tapers for medical use, medical tubing for IV (intravenous) access and infusion, drug infusion adapters for connecting infusion fluid containers with syringes, implantable medical fluid injection ports, implantable medical valve ports, catheters, cannulas in the nature of medical tubing for drainage, transfusion, and administering drugs, medical adapters in the nature of Luer tapers for medical use, needles for medical use, medical syringes, medical tubing for IV (intravenous) access and infusion, medical fluid flow control devices in the nature of IV (intravenous) access tubing and general IV (intravenous) access tubing for administering drugs and fluids; and parts and fittings for the foregoing goods

Goods & Services
Medical devices and apparatus, namely, needleless access devices in the nature of needleless vascular access ports for medical use, needleless blood drawing devices, blood collection needles and tubes, and blood drawing devices; phlebotomy devices, namely, blood drawing tubes, needles, winged needle sets comprised of winged blood collection needles and blood collecting tubes, syringes for medical use; needleless injection devices for pharmaceuticals or IV (intravenous) fluids for medical use, needleless adapters in the nature of Luer tapers for medical use, needleless connectors in the nature of Luer tapers for medical use, medical fluid connectors in the nature of Luer tapers for medical use, drug infusion connectors in the nature of Luer tapers for medical use, medical tubing for IV (intravenous) access and infusion, drug infusion adapters for connecting infusion fluid containers with syringes, implantable medical fluid injection ports, implantable medical valve ports, catheters, cannulas in the nature of medical tubing for drainage, transfusion, and administering drugs, medical adapters in the nature of Luer tapers for medical use, needles for medical use, medical syringes, medical tubing for IV (intravenous) access and infusion, medical fluid flow control devices in the nature of IV (intravenous) access tubing and general IV (intravenous) access tubing for administering drugs and fluids; and parts and fittings for the foregoing goods

Goods & Services
Medical devices and apparatus, namely, catheters, needles for medical use, needle-free and needleless infusion injection ports; medical devices for stabilizing peripheral IV catheters; IV (intravenous) extension sets comprised of luer tapers for medical use, IV catheter extension tubing, and medical devices for stabilizing IV catheters for delivering intravenous fluids from an IV bag or from a syringe to a patient; closed IV catheter sets comprised of luer tapers for medical use, IV catheter extension tubing, and medical devices for stabilizing IV catheters for delivering intravenous fluids from an IV bag or from a syringe to a patient; infusion devices for administering drugs

Goods & Services
Downloadable software in the nature of a mobile application for controlling and managing patient medical information; downloadable software in the nature of a mobile application for the collection, editing, organizing, modifying, transmission, storage and sharing of patient care instruction and medical information; Providing temporary use of non-downloadable cloud-based software for controlling and managing patient medical information; providing temporary use of non-downloadable cloud-based software for the collection, editing, organizing, modifying, transmission, storage and sharing of patient care instruction and medical information

Goods & Services
Medical specimen collection, transfer, and dispensing devices in the nature of tubes for the collection, transferring, and dispensing of blood specimens


Goods & Services
Product development support program in the nature of development and regulatory submission management, namely, providing business assistance to others in developing products and preparing and filing applications for combination products with governmental regulatory bodies
Goods & Services
Laboratory flow cytometer

Goods & Services
Laboratory cell analyzer, namely, flow cytometers; cell separator, namely, flow cytometers

Goods & Services
Reagents for scientific use

Goods & Services
Reagents for scientific use

Goods & Services
Chemical preparations for scientific purposes; cell culture media supplements for laboratory use, scientific and research use; chemical, biological and diagnostic reagents and prepared laboratory media for scientific or research purposes; chemicals for flow cytometry for non-medical purposes, for use in industry and science; fluorescent dyes for scientific laboratory and research use; biological preparation for use in hamster ovary cell culture media other than for medical or veterinary use; molecular testing reagents for scientific or research use; cell culture media for in vitro diagnostic laboratory testing; cell culture media supplement used for the production of biological therapeutics; research reagents for research laboratory use, namely, flow cytometry single-vial reagents; flow cytometry kits comprised of research reagents, buffers and controls for research laboratory use; stem cell culture reagent system comprising chemical culture nutrients, culture growth proteins and protein microarrays for the expansion of mesenchymal stem cells for research and manufacturing use; molecular assays for the detection and identification of infectious diseases for research purposes; laboratory reagents for in vitro diagnostic use in infectious disease testing; reagents for scientific or medical research use, namely, for preparing blood samples for antibody staining in immunophenotyping; chemically impregnated strips and discs for in vitro diagnosis of various diseases; chemical aerosol spray fixative for cytological smears carried on a slide; test kits for detection of antibodies to rubella virus comprising essentially an agglutination card, a latex reagent, serum and a buffer solution for laboratory or research use; disposable packet containing chemicals for generating hydrogen gas and carbon dioxide for scientific purposes; anti-microbial products, namely, chemical test kits for laboratory use in testing body fluid specimens for laboratory or research use; clinical or medical laboratory in vitro diagnostic preparation for the identification of microorganisms | Chemical and diagnostic preparations for medical purposes for treatment of infectious diseases; pharmaceutical preparations for the treatment and prevention of infectious diseases; diagnostic test kits featuring antibodies and reagents for the detection of viral and bacterial infections and infectious diseases; sanitizing wipes; chemical, biological and diagnostic reagents and prepared laboratory media for clinical or medical purposes; prefilled syringes containing saline and heparin; chemical reagents for medical purposes for flow cytometry; medical diagnostic assays for the detection of infectious diseases; medical kits comprising a syringe prefilled with saline or heparinized saline and a disinfectant prep pad; in vitro diagnostic reagents for medical use, and assay kits for medical use comprising medical diagnostic reagents for the detection of diseases including cancer and HPV; molecular testing reagents for medical use; medical diagnostic molecular assays for the detection and identification of infectious diseases; media for bacteriological laboratory cultures; surgical scrub, namely, antimicrobial surgical scrubbing solution; medical dressings; sterile sanitizing wipes; povidone iodine swabs and preparations for medical purposes; pharmaceutical preparation consisting essentially of a mixed culture of lactobacillus acidophilus and lactobacillus bulgaricus for oral administration for the treatment of intestinal disorders; broad-spectrum antiseptic; antimicrobial catheter patch medical dressings; medical liquid applicators containing antimicrobial solution and used for applying antimicrobial solution; antiseptic cotton swabs for medical use; adhesive medical bandages for keeping intravenous needles and luer fittings in place; Nonwoven sterilization preparation wrap used in connection with surgical instrument set, namely, medical dressings; medical dressing, namely, sterile wraps for use in connection with medical procedures such as in surgical operations | Computer hardware for scientific purposes; laboratory instruments, namely, assay plates, microplates, coverslips, petri dishes, pipets, tubes, specimen transport containers, anaerobic jars and coagulation timers, flow cytometers and downloadable computer operating software therefor; laboratory apparatus for storing and testing blood cultures; scientific instruments, namely, sample preparation and real-time polymerase chain reaction (RT-PCR) analyzer for the detection and identification of infectious diseases for scientific use; Downloadable computer software and computer hardware for the preparation of medications; Downloadable computer software for the preparation of pharmaceuticals; Downloadable computer software for counting, sorting and detecting cells; computer hardware and downloadable software used for remote diagnostics and troubleshooting of laboratory instruments; Downloadable computer software for use in infectious disease management and healthcare worker safety; Downloadable computer programs and program manuals all sold to the user as a unit for use by hospitals and health related groups for tracking employee medical histories; Downloadable computer programs and manuals for use by medical facilities in monitoring and controlling infections; Downloadable computer software used for diabetes management; Downloadable computer software used for a patient identification bar code system; laboratory instrument accessory, namely, an antimicrobial susceptibility test touch panel for the automated detection of pathogens in the presence of antimicrobials in a biological sample for research use; electronic laboratory apparatus, namely, clinical specimen processor and biochemical analyzer used for clinical and laboratory research and in vitro clinical diagnostics; downloadable electronic technical newsletters in the field of phlebotomy and diagnostic testing provided via a website; laboratory instruments for hematology determination; temperature controlled test tube incubator for use in blood banks and hematology laboratories or laboratory use; electronic laboratory apparatus, namely, a sample processor used for clinical and laboratory research and clinical diagnostics; laboratory apparatus, namely, slide and blood culture bottle used for subculturing microorganisms from liquid growth media; Laboratory diagnostic instrument, namely, a robotic processor which performs DNA probe hybridization and color development to differentiate and identify vaginitis pathogens for laboratory use; laboratory instruments and apparatus, namely, plates for the isolation of disease-causing microorganisms, lids used to cover the plates and maintain isolation from the environment, pipettes, dishes for containing culture media, inserts for cell culture growth and for performing cell-based assays, laboratory plastic ware, namely, tissue culture flasks, roller bottles, cell culture dishes and plates, petri dishes, conical and round-bottom tubes, pipettes, containers, specifically test tubes, trays for holding test cards, laboratory filters, laboratory glassware, namely, bottles, cap covers used to seal specimen transport containers and tubed culture media, culture slides, cell scrapers, cell strainers, cellware, namely, plastic multi-well plates, flasks, vials, bottles, scrapers, inserts, dishes, slides, tubes and pipettes for laboratory use, high throughput screening kits consisting of laboratory pipettes, reagents, tissues, cells, tubes, vials, flasks, bottles, multi-well plates, slides and flasks, sold as a unit for scientific and research use; laboratory apparatus, namely, test cards for in vitro diagnostic testing of samples for infectious diseases; laboratory apparatus, namely, incubator and laboratory card rotator therefor for use in infectious disease testing; automated machines for dispensing dosages of medicine and medical supplies; computer hardware and downloadable computer software for medication order management, and for securing, tracking, monitoring and replenishment of medicine and medical supplies for medicine and medical supply dispensing machines; computer controlled system comprised of an integrated dispenser unit and a separate dispensing and return unit, that electronically dispenses and tracks scrub suits and other garments used in medical facilities to authorized employees and then receives used scrubs for clean replacements; automated supply management system comprised of bar code scanners, downloadable software and a central computer for verifying the selection and replenishment of supplies and other items into supply storage areas; downloadable computer software for operating a computer controlled system comprised of electronic automated machines for dispensing medical supplies, for identifying patients, procedures and physicians, for managing supply costs, for creating and editing kits designed for particular medical procedures and for tracking the usage of items dispensed by the machines; downloadable computer software used for performing medical diagnostics and management of patient data, and for data management, organization of patient data, workflow management and connectivity in the nature of maintaining communication between patients and healthcare providers, all of the foregoing for use in the medical field; electronic control devices and apparatus for storing and removal of individually packaged goods or wares, in particular pharmaceuticals; downloadable computer operating programs used for controlling devices and apparatus which are used for storing and removal of individually packaged goods or wares; electronic control systems comprising computer hardware and downloadable computer software for machinery used for storing and removal of individually packaged goods and wares, in particular pharmaceuticals; data processing equipment, namely, computer hardware and downloadable data processing software, for medical purposes; automated dispensing machines featuring a computer, downloadable computer software and printer all sold as a unit for dispensing and packing prescription medications; downloadable computer software that integrates patients, costs, treatments and outcome of medical data of healthcare institutions; secured medication dispensing machines and inventory management systems comprising of computer hardware and downloadable software for maintaining the secured medication dispensing machines and inventory management systems; downloadable software to integrate physiological monitoring devices, namely, devices that monitor the normal functions of living organisms and their parts and instructional manuals sold as a unit; downloadable software for analyzing physiological data, namely, software that monitors the normal functions of living organisms and their parts and instructional manuals sold as a unit; a handheld data input capturing tool for use in the medical field, especially for clinical trials and disease management, comprising of a writing stylus and an optical input, wherein the optical input reads information written by the writing stylus and records said information in its electronic memory, and wherein the device is capable of transmitting said recorded information to other electronic devices, namely, computers, computer memory, computer databases, magnetic storage media, optical storage media, by direct communication, or modem, or internet; automated medication management system comprised of bar code scanners, label printers, and a computer interface, for verifying the selection and replenishment of media and other items into medication dispensing machines; downloadable computer programs for storing medication delivery parameters, namely, drug names, drug conflicts, flow rates, dosages, volumes, delivery time periods, patient physiological parameters, vital signs parameters, and monitoring medication delivery parameters selected by a care giver, and comparing the medication delivery parameters selected by a care giver to stored medication delivery parameters, and providing display data based on the comparison; fluid flow rate meters; ammeters; downloadable computer programs for controlling and monitoring fluid flows and detecting obstructions to fluid flows in the medical industry and downloadable computer programs for capturing, storing, integrating, and presenting data in patient care management systems; downloadable medical software for use in cell sorting instruments for flow cytometry; laboratory ovens, featuring thermal regulators and thermostats; laboratory centrifuges; medical laboratory apparatus for promoting controlled tissue growth, namely, laboratory incubators, laboratory climatic test cabinets, laboratory shakers, medical instruments for measuring temperature humidity and pH-value, all for use in biotechnology and microbiology | Medical instruments and apparatus, namely, syringes, stoppers for syringes, syringe holders, needle holders, hypodermic needles, hypodermic needle injectors, catheters, sample collection devices in the nature of specimen cups and tubes, centrifuges, lancets, surgical blades, handles for surgical instruments and apparatus, surgical drapes, surgical scrub brushes, surgical sponges, tubes for surgical instruments and apparatus, tubing for surgical instruments and apparatus, stopcocks for surgical instruments and apparatus, adapters for surgical instruments and apparatus, holders for surgical instruments and apparatus, access valves for surgical instruments and apparatus, extension sets featuring tubing, valves and connectors, cannulae and medical guidewires; medical transducer kits comprised of drip chambers, tubing for use with medical devices, stopcocks valves, pressure transducers, access ports, and male and female luer adapters, all for use in administering medical fluids; medical infusion sets consisting of drip chamber, tubing, roller clamp, access ports and male luer adapter for use in administering medical fluids; containers for medical waste; Medical devices, namely, blood analyzers and cell separators, sorters and counters for medical diagnosis; medical instruments and apparatus, namely, tubes for drainage, transfusion, and administering drugs; medical instruments and apparatus, namely, tubing for infusion of fluids, administration of drugs; medical instruments and apparatus, namely, for blood sample collection and for drainage; medical instruments and apparatus, namely, stopcocks for infusion of fluids, administration of drugs, blood sample collection and for drainage; medical instruments and apparatus, namely, holders for blood collection tubes for drainage and specimen collection; medical instruments and apparatus, namely, access valves for drainage, transfusion, administering drugs and specimen collection; medical instruments and apparatus, namely, extension sets for infusion of fluids, administration of drugs, blood sample collection and for drainage; medical instruments and apparatus, namely, infusion sets, and containers for medical waste and replacement parts therefor being sold as a unit therewith; inhalers sold empty for nasal drug delivery; medical devices, namely, IV sets comprising of empty IV medicament solution bags, tubing, IV line access valves, catheters, extension sets for fluid delivery; flow cytometers for clinical purposes for medical diagnostic uses; needle shielding system comprised of a protective sleeve and needle cover for use with a prefillable syringe; drug delivery systems, namely, patch injectors, patch pumps and microinfusors; medical kits for diabetic patients comprised primarily of syringes, pen needles, lancets, glucose tablets, educational materials and disinfectant prep pads; injection and infusion hypodermic needles used to deliver medications to manage diabetes; automated medical processing system for screening cervical cancer and sexually transmitted diseases comprised of an instrument for receiving samples of specimens, an medical instrument for preparing specimen slides, medical imaging apparatus and an instrument for molecular testing for infections; medical injection systems comprising of syringes and medical instruments for injections; sample preparation device for medical diagnostic use; medical apparatus and instruments, namely, scale magnifier and needle guide for insulin syringes; medical diagnostic instrument, namely, a robotic processor which performs DNA probe hybridization and color development to differentiate and identify vaginitis pathogens for medical use; single limb anesthesia breathing circuit machines for use in patient care; medical apparatus and instruments, namely, needleless intravenous connector for medical fluids; medical apparatus and instruments, namely, bone cement delivery system comprised of medical apparatus and instruments for the mixing, application and injection of bone cement and radiopaque bone cement; biopsy hypodermic needles; surgical stapler; medical devices, namely, infusion pumps for delivering measured amounts of solutions into the bloodstream over time; medical apparatus and instruments, namely, medical fluid connectors for use in the administration of medical fluids, drug infusion connectors for use in the administration of medical fluids, medical infusion bags for use in storing medical fluids for administration, medical vial access adapters for use in piercing septa of closed medical vials to permit access to the vial's contents; anterior cervical fusion retractor used in surgery of the spine; lumbar spine retractor used for surgery of the spine; medical devices, namely, bone punchers and snippers for use in surgery; rongeurs for use in surgery; kerrisons for use in surgery; handheld surgical instruments for neurosurgery and cadaver head holders for use in surgical procedures; laparoscopic surgical instruments, namely, ratchets; insertable and implantable medical devices made from artificial materials, namely, shunts and valves; doppler ultrasound medical device used to aid in the detection of peripheral vascular disease and to listen to blood flow in the fetal heart; medical device, namely, tongue positioning mouthpiece; electro-medical devices used to administer and monitor anaesthetics, anaesthetic gases and oxygen, and structural parts for such apparatus; cardiovascular medical apparatuses, namely, apparatus for measuring arterial stiffness in a person, and apparatus for measuring arterial elasticity and endothelial function in a person; clinical vital sign medical measurement instruments; medical apparatus, namely, fiberoptic light system comprising of an adjustable headlight band with an attached fiber optic cable for providing light from a light source to be used in medical and surgical procedures; medical apparatus, namely, preoperative hair removal clippers and surgical blades for using during surgical procedures; medical devices, namely, needleless intravenous connector for medical fluids; medical devices, namely, molded tubing apparatus for medical use, namely, universal connectors and protective caps, needleless connectors, and intravenous manifold connectors for use in medical fluid control; medical devices that allow intravenous access via a syringe or male luer; medical instruments that monitor the delivery of medication to a patient, that store medication delivery parameters, namely, drug names, drug conflicts, flow rates, dosages, volumes, delivery time periods, patient physiological parameters, vital signs parameters, and that monitor medication delivery parameters selected by a care giver, and that compare the medication delivery parameters selected by a care giver to stored medication delivery parameters, and that provide display data based on the comparison, namely, medication delivery monitors, infusion pumps, syringe pumps, programmable infusion pumps, programmable syringe pumps and the resident control computer programs sold as an integral component thereof; surgical instruments, namely, blade tips, shafts, scissors rasps, and osteotomes; surgical clamps and forceps; thermometers, disposable thermometer covers, thermometer probes, thermometer stands, thermometer probe cover holders, thermometer base units, all for medical purposes; medical device, namely, medical compression stockings and tights; biopsy hypodermic needle for surgical and medical use; medical apparatus, namely, medical fluid conducting apparatus for use with medical infusion pumps; surgical instruments, namely, retractors, graspers and dissectors; medical devices, namely, volumetric pump and controller devices used as part of an administration set for the infusion of medical solutions to patients; surgical instruments; endoscopic telescopes for medical purposes; blood pressure measuring apparatus, namely, disposable blood pressure cuffs; medical instruments for measuring and monitoring blood pressure, pulse and temperature; surgical instruments for cutting, retracting, grasping, holding, occluding, dilating, probing, cannulating, draining, aspirating, suturing and ligating in the various fields of surgical practice, namely, surgical knives, scissors, medical chisels, osteotomes, curettes, dissectors, forcep and periosteal elevators, rongeurs, cutting forceps and punches, medical saws, trocars, biopsy hypodermic needles, medical retractors, both hand held and self-retaining, tenacula and vulsellum forceps, hemostatic artery forceps, ansatomosis clamps, dressing and tissue forceps, needle holding forceps, lacrimal probes for medical purposes, sinus probes, gall duct probes, urethral, ureteral, uterine and mitral valve dilators for medical purposes, suction tubes and cannulas, suture needles and ligating instruments for general surgery, rectal and colon procedures, plastic and reconstructive surgery, thoracic and cardiovascular surgery, gynecological and obstetrical procedures, urologic surgery, microsurgery, ophthalmic surgery, ear and endaural surgery, nasal sinus and reconstruction procedures, oral, tonsil, laryngeal, bronchoscopic and esophagoscopic procedures, pediatric surgery, neurological surgery and orthopaedic surgery; suture hypodermic needles and ligating medical instruments; biopsy hypodermic needles, punches and forceps; anesthesia equipment, namely, medical suction tubes, airways, tube-holding forceps; medical electronic instruments, namely, electronic ocular tonometers, and tonographers; diagnostic endoscopes for medical use; surgical supplies, namely, drapes for surgical procedures; surgical equipment, namely, a tray and basin system for disinfecting, transporting and storage of surgical instruments; tourniquet inflation units, namely, medical devices for use in cannulation or to stem the flow of traumatic bleeding, namely, tourniquets; patient examining and treatment tables; surgical supplies, namely, drapes for surgical procedures; surgical equipment, namely, a tray and basin system for disinfecting; medical tubes, vials and envelopes used for transporting fluid bacteriological specimens, for medical purposes, sold empty; medical devices, namely, infusion pumps for delivering measured amounts of solutions into the bloodstream over time; medical diagnostic instruments for the analysis of body fluid; medical devices for obtaining body fluid samples for laboratory use in testing body fluid specimens | Education and training services, namely, classes, seminars and workshops in the fields of medicine, medical research, medical analysis, medical diagnosis, and medical product development; educational services, namely, conducting lectures, workshops, web-based seminars and roundtable discussions focused on key industry trends in healthcare for professionals; training in the operation, repair and maintenance of diagnostic instruments, reagents and software for use with such instruments in the fields of life science research, pharmaceutical development, clinical care and biotechnology; providing online newsletters in the field of diabetes healthcare; educational services, namely, providing online instruction in the fields of medical technology, diabetes and diabetes care; educational services, namely, professional training in the field of diabetes healthcare, medical specimen collection; education services, namely, providing seminars and on-line seminars in the fields of medical technology, safe medical injection and diabetes care, and distributing print and electronic course materials in connection therewith; educational services, namely, providing training in the field of diabetes and diabetes care provided by means of a call center and distribution of printed and electronic training materials in connection therewith; providing education services in the field of women's health and cancer in the form of live and on-line training, classes and seminars; educational services, namely, providing continuing professional education program, namely, lectures, workshops and roundtable discussions focused on medical sample collection, processing and transport, the preanalytical phase of laboratory testing, troubleshooting incorrect handling procedures, and disease etiology; educational services, namely, in-service training programs to educate customers on compliance with the healthcare safety acts designed for healthcare workers who may be exposed to disease while handling certain medical devices, to educate customers on proper use of safety-engineered products, and assist customers with conversions on how to use safety-engineered products; clinical education services, namely, providing mentoring, tutoring, classes, seminars and workshops in the fields of oncology, interventional procedures, malignant pleural effusions, and vertebral augmentation; medical training and teaching

Goods & Services
Medical devices and apparatus, comprised of an ultrasound machine, ultrasound probes, ultrasound probe covers, recorded medical imaging software, an activator for magnetizing the needle, and also including electrical power supply, roll stands especially adapted for mounting medical devices and replacement parts and fittings for the ultrasound machine
Goods & Services
Molecular assays for the detection and identification of infectious diseases for research purposes | Medical diagnostic molecular assays for the detection and identification of infectious diseases | Laboratory equipment, namely, sample preparation and real-time polymerase chain reaction (RT-PCR) analyzer for the detection and identification of infectious diseases for scientific use
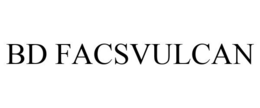
Goods & Services
Imaging flow cytometer for general research use

Goods & Services
Reagents sold in kit form for in vitro diagnostic use; molecular assays for the detection and identification of infectious diseases for research purposes | Clinical or medical laboratory in vitro diagnostic preparation for the identification of microorganisms; Medical diagnostic molecular assays for the detection and identification of infectious diseases | Specimen containers used for the collection and transport of specimens to the microbiology laboratory for organism recovery; petri dishes, anaerobic jars and coagulation timers; laboratory analytical apparatus, namely, an analyzer for the identification and/or antimicrobial susceptibility testing of microbes; Laboratory apparatus, namely, a sample processor used for clinical and laboratory research and clinical diagnostics; Device for the preparation of cytology thin-layer slides for scientific use; Automated laboratory robot and related recorded operating software, which prepares liquid-based, thin layer cellular samples from a cell suspension for use in medical, clinical and scientific procedures and devices for use therewith, namely, vials, tubes, disposable lab testing kits, pipettes, settling chambers and slides, sold as a unit therewith; Laboratory equipment, namely, sample preparation and real-time polymerase chain reaction (RT-PCR) analyzer for the detection and identification of infectious diseases for scientific use; apparatus for storing and testing blood cultures; laboratory apparatus and instruments for inoculation, processing and testing of microbiological samples; computer hardware and downloadable software for use in conventional and thin layer pap smear image diagnostics; computer hardware and downloadable software for diagnostic imaging using automated cell location; computer hardware and downloadable software for use in data and image processing in the field of computer-aided medical microscopy | Medical apparatus, namely, a device for diagnostic imaging of conventional and thin layer pap smears; video cameras for use in the field of computer-aided medical microscopy; manual and automated microscopes for use in the field of computer-aided medical microscopy; Device for the preparation of cytology thin-layer slides for medical use and for clinical medical use


Goods & Services
Topical antimicrobial solutions; pre-surgical antibacterial skin preparations
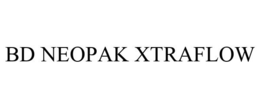



Goods & Services
Medical instruments, namely, IV catheters and cannula

Owner:
Serial:88590645
Filed:Aug 23, 2019
Classes:1, 5, 9, +2
Registration:6134144
Registered:Aug 25, 2020
Goods & Services
Chemical preparations for scientific purposes; cell culture media supplements for laboratory use, scientific and research use; chemical, biological and diagnostic reagents and prepared laboratory media for scientific or research purposes; chemicals for flow cytometry for non-medical purposes, for use in industry and science; fluorescent dyes for scientific laboratory and research use; biological preparation for use in hamster ovary cell culture media other than for medical or veterinary use; molecular testing reagents for scientific or research use; cell culture media for in vitro diagnostic laboratory testing; cell culture media supplement used for the production of biological therapeutics; research reagents for research laboratory use, namely, flow cytometry single-vial reagents; flow cytometry kits comprised of research reagents, buffers and controls for research laboratory use; stem cell culture reagent system comprising chemical culture nutrients, culture growth proteins and protein microarrays for the expansion of mesenchymal stem cells for research and manufacturing use; molecular assays for the detection and identification of infectious diseases for research purposes; laboratory reagents for in vitro diagnostic use in infectious disease testing; reagents for scientific or medical research use, namely, for preparing blood samples for antibody staining in immunophenotyping; chemically impregnated strips and discs for in vitro diagnosis of various diseases; chemical aerosol spray fixative for cytological smears carried on a slide; test kits for detection of antibodies to rubella virus comprising essentially an agglutination card, a latex reagent, serum and a buffer solution for laboratory or research use; disposable packet containing chemicals for generating hydrogen gas and carbon dioxide for scientific purposes; anti-microbial products, namely, chemical test kits for laboratory use in testing body fluid specimens for laboratory or research use | Chemical and diagnostic preparations for medical purposes for treatment of infectious diseases; pharmaceutical preparations for the treatment and prevention of infectious diseases; diagnostic test kits featuring antibodies and reagents for the detection of viral and bacterial infections and infectious diseases; sanitizing wipes; chemical, biological and diagnostic reagents and prepared laboratory media for clinical or medical purposes; prefilled syringes containing saline and heparin; chemical reagents for medical purposes for flow cytometry; medical diagnostic assays for the detection of infectious diseases; medical kits comprising a syringe prefilled with saline or heparinized saline and a disinfectant prep pad; in vitro diagnostic reagents for medical use, and assay kits for medical use comprising medical diagnostic reagents for the detection of diseases including cancer and HPV; molecular testing reagents for medical use; medical diagnostic molecular assays for the detection and identification of infectious diseases; media for bacteriological laboratory cultures; surgical scrub, namely, antimicrobial surgical scrubbing solution; medical dressings; sterile sanitizing wipes; povidone iodine swabs and preparations for medical purposes; clinical or medical laboratory in vitro diagnostic preparation for the identification of microorganisms; pharmaceutical preparation consisting essentially of a mixed culture of lactobacillus acidophilus and lactobacillus bulgaricus for oral administration for the treatment of intestinal disorders; broad-spectrum antiseptic; antimicrobial catheter patch medical dressings; medical liquid applicators containing antimicrobial solution and used for applying antimicrobial solution; antiseptic cotton swabs for medical use; adhesive medical bandages for keeping intravenous needles and luer fittings in place; Nonwoven sterilization preparation wrap used in connection with surgical instrument set, namely, medical dressings | Computer hardware for scientific purposes; laboratory instruments, namely, assay plates, microplates, coverslips, petri dishes, pipets, tubes, specimen transport containers, anaerobic jars and coagulation timers, flow cytometers and downloadable computer operating software therefor; laboratory apparatus for storing and testing blood cultures; scientific instruments, namely, sample preparation and real-time polymerase chain reaction (RT-PCR) analyzer for the detection and identification of infectious diseases for scientific use; Downloadable computer software and computer hardware for the preparation of medications; Downloadable computer software for the preparation of pharmaceuticals; Downloadable computer software for counting, sorting and detecting cells; computer hardware and downloadable software used for remote diagnostics and troubleshooting of laboratory instruments; Downloadable computer software for use in infectious disease management and healthcare worker safety; Downloadable computer programs and program manuals all sold to the user as a unit for use by hospitals and health related groups for tracking employee medical histories; Downloadable computer programs and manuals for use by medical facilities in monitoring and controlling infections; Downloadable computer software used for diabetes management; Downloadable computer software used for a patient identification bar code system; laboratory instrument accessory, namely, an antimicrobial susceptibility test touch panel for the automated detection of pathogens in the presence of antimicrobials in a biological sample for research use; electronic laboratory apparatus, namely, clinical specimen processor and biochemical analyzer used for clinical and laboratory research and in vitro clinical diagnostics; downloadable electronic technical newsletters in the field of phlebotomy and diagnostic testing provided via a website; laboratory instruments for hematology determination; temperature controlled test tube incubator for use in blood banks and hematology laboratories or laboratory use; electronic laboratory apparatus, namely, a sample processor used for clinical and laboratory research and clinical diagnostics; laboratory apparatus, namely, slide and blood culture bottle used for subculturing microorganisms from liquid growth media; Laboratory diagnostic instrument, namely, a robotic processor which performs DNA probe hybridization and color development to differentiate and identify vaginitis pathogens for laboratory use; laboratory instruments and apparatus, namely, plates for the isolation of disease-causing microorganisms, lids used to cover the plates and maintain isolation from the environment, pipettes, dishes for containing culture media, inserts for cell culture growth and for performing cell-based assays, laboratory plastic ware, namely, tissue culture flasks, roller bottles, cell culture dishes and plates, petri dishes, conical and round-bottom tubes, pipettes, containers, specifically test tubes, trays for holding test cards, laboratory filters, laboratory glassware, namely, bottles, cap covers used to seal specimen transport containers and tubed culture media, culture slides, cell scrapers, cell strainers, cellware, namely, plastic multi-well plates, flasks, vials, bottles, scrapers, inserts, dishes, slides, tubes and pipettes for laboratory use, high throughput screening kits consisting of laboratory pipettes, reagents, tissues, cells, tubes, vials, flasks, bottles, multi-well plates, slides and flasks, sold as a unit for scientific and research use; laboratory apparatus, namely, test cards for in vitro diagnostic testing of samples for infectious diseases; laboratory apparatus, namely, incubator and laboratory card rotator therefor for use in infectious disease testing; automated machines for dispensing dosages of medicine and medical supplies; computer hardware and downloadable computer software for medication order management, and for securing, tracking, monitoring and replenishment of medicine and medical supplies for medicine and medical supply dispensing machines; computer controlled system comprised of an integrated dispenser unit and a separate dispensing and return unit, that electronically dispenses and tracks scrub suits and other garments used in medical facilities to authorized employees and then receives used scrubs for clean replacements; automated supply management system comprised of bar code scanners, downloadable software and a central computer for verifying the selection and replenishment of supplies and other items into supply storage areas; downloadable computer software for operating a computer controlled system comprised of electronic automated machines for dispensing medical supplies, for identifying patients, procedures and physicians, for managing supply costs, for creating and editing kits designed for particular medical procedures and for tracking the usage of items dispensed by the machines; downloadable computer software used for performing medical diagnostics and management of patient data, and for data management, organization of patient data, workflow management and connectivity in the nature of maintaining communication between patients and healthcare providers, all of the foregoing for use in the medical field; electronic control devices and apparatus for storing and removal of individually packaged goods or wares, in particular pharmaceuticals; downloadable computer operating programs used for controlling devices and apparatus which are used for storing and removal of individually packaged goods or wares; electronic control systems comprising computer hardware and downloadable computer software for machinery used for storing and removal of individually packaged goods and wares, in particular pharmaceuticals; data processing equipment, namely, computer hardware and downloadable data processing software, for medical purposes; automated dispensing machines featuring a computer, downloadable computer software and printer all sold as a unit for dispensing and packing prescription medications; downloadable computer software that integrates patients, costs, treatments and outcome of medical data of healthcare institutions; secured medication dispensing machines and inventory management systems comprising of computer hardware and downloadable software for maintaining the secured medication dispensing machines and inventory management systems; downloadable software to integrate physiological monitoring devices, namely, devices that monitor the normal functions of living organisms and their parts and instructional manuals sold as a unit; downloadable software for analyzing physiological data, namely, software that monitors the normal functions of living organisms and their parts and instructional manuals sold as a unit; a handheld data input capturing tool for use in the medical field, especially for clinical trials and disease management, comprising of a writing stylus and an optical input, wherein the optical input reads information written by the writing stylus and records said information in its electronic memory, and wherein the device is capable of transmitting said recorded information to other electronic devices, namely, computers, computer memory, computer databases, magnetic storage media, optical storage media, by direct communication, or modem, or internet; automated medication management system comprised of bar code scanners, label printers, and a computer interface, for verifying the selection and replenishment of media and other items into medication dispensing machines; downloadable computer programs for storing medication delivery parameters, namely, drug names, drug conflicts, flow rates, dosages, volumes, delivery time periods, patient physiological parameters, vital signs parameters, and monitoring medication delivery parameters selected by a care giver, and comparing the medication delivery parameters selected by a care giver to stored medication delivery parameters, and providing display data based on the comparison; fluid flow rate meters; ammeters; downloadable computer programs for controlling and monitoring fluid flows and detecting obstructions to fluid flows in the medical industry and downloadable computer programs for capturing, storing, integrating, and presenting data in patient care management systems; downloadable medical software for use in cell sorting instruments for flow cytometry; laboratory ovens, featuring thermal regulators and thermostats; laboratory centrifuges; medical dressing, namely, sterile wraps for use in connection with medical procedures such as in surgical operations | Medical instruments and apparatus, namely, syringes, stoppers for syringes, syringe holders, needle holders, hypodermic needles, hypodermic needle injectors, catheters, sample collection devices in the nature of specimen cups and tubes, centrifuges, lancets, surgical blades, handles for surgical instruments and apparatus, surgical drapes, surgical scrub brushes, surgical sponges, tubes for surgical instruments and apparatus, tubing for surgical instruments and apparatus, stopcocks for surgical instruments and apparatus, adapters for surgical instruments and apparatus, holders for surgical instruments and apparatus, access valves for surgical instruments and apparatus, extension sets featuring tubing, valves and connectors for drawing blood and intravenous administration of fluids; cannulae and medical guidewires; medical transducer kits comprised of drip chambers, tubing for use with medical devices, stopcocks valves, pressure transducers, access ports, and male and female luer adapters, all for use in administering medical fluids; medical infusion sets consisting of drip chamber, tubing, roller clamp, access ports and male luer adapter for use in administering medical fluids; containers for medical waste; Medical devices, namely, blood analyzers and cell separators, sorters and counters for medical diagnosis; medical instruments and apparatus, namely, tubes for drainage, transfusion, and administering drugs; medical instruments and apparatus, namely, tubing for infusion of fluids, administration of drugs; medical instruments and apparatus, namely, for blood sample collection and for drainage; medical instruments and apparatus, namely, stopcocks for infusion of fluids, administration of drugs, blood sample collection and for drainage; medical instruments and apparatus, namely, holders for blood collection tubes for drainage and specimen collection; medical instruments and apparatus, namely, access valves for drainage, transfusion, administering drugs and specimen collection; medical instruments and apparatus, namely, extension sets for infusion of fluids, administration of drugs, blood sample collection and for drainage; medical instruments and apparatus, namely, infusion sets, and containers for medical waste and replacement parts therefor being sold as a unit therewith; inhalers sold empty for nasal drug delivery; medical devices, namely, IV sets comprising of empty IV medicament solution bags, tubing, IV line access valves, catheters, extension sets for fluid delivery; flow cytometers for clinical purposes for medical diagnostic uses; needle shielding system comprised of a protective sleeve and needle cover for use with a prefillable syringe; drug delivery systems, namely, patch injectors, patch pumps and microinfusors; medical kits for diabetic patients comprised primarily of syringes, pen needles, lancets, glucose tablets, educational materials and disinfectant prep pads; injection and infusion hypodermic needles used to deliver medications to manage diabetes; automated medical processing system for screening cervical cancer and sexually transmitted diseases comprised of an instrument for receiving samples of specimens, an medical instrument for preparing specimen slides, medical imaging apparatus and an instrument for molecular testing for infections; medical injection systems comprising of syringes and medical instruments for injections; sample preparation device for medical diagnostic use; medical apparatus and instruments, namely, scale magnifier and needle guide for insulin syringes; medial diagnostic instrument, namely, a robotic processor which performs DNA probe hybridization and color development to differentiate and identify vaginitis pathogens for medical use; medical apparatus for promoting controlled tissue growth, namely, laboratory incubators, laboratory climatic test cabinets, laboratory shakers, medical instruments for measuring temperature humidity and pH-value, all for use in biotechnology and microbiology; medial diagnostic instrument, namely, a robotic processor which performs DNA probe hybridization and color development to differentiate and identify vaginitis pathogens for medical use; single limb anesthesia breathing circuit machines for use in patient care; medical apparatus and instruments, namely, needleless intravenous connector for medical fluids; medical apparatus and instruments, namely, bone cement delivery system comprised of medical apparatus and instruments for the mixing, application and injection of bone cement and radiopaque bone cement; biopsy hypodermic needles; surgical stapler; medical devices, namely, infusion pumps for delivering measured amounts of solutions into the bloodstream over time; medical apparatus and instruments, namely, medical fluid connectors for use in the administration of medical fluids, drug infusion connectors for use in the administration of medical fluids, medical infusion bags for use in storing medical fluids for administration, medical vial access adapters for use in piercing septa of closed medical vials to permit access to the vial's contents; anterior cervical fusion retractor used in surgery of the spine; lumbar spine retractor used for surgery of the spine; medical devices, namely, bone punchers and snippers for use in surgery; rongeurs for use in surgery; kerrisons for use in surgery; handheld surgical instruments for neurosurgery and cadaver head holders for use in surgical procedures; laparoscopic surgical instruments, namely, ratchets; insertable and implantable medical devices made from artificial materials, namely, shunts and valves; doppler ultrasound medical device used to aid in the detection of peripheral vascular disease and to listen to blood flow in the fetal heart; medical device, namely, tongue positioning mouthpiece; electro-medical devices used to administer and monitor anaesthetics, anaesthetic gases and oxygen, and structural parts for such apparatus; sterilization containers, baskets, trays, cassettes and accessories therefor, namely, racks for holding medical instruments and metal identification tags for medical instruments, all sold as a unit and all for medical use; cardiovascular medical apparatuses, namely, apparatus for measuring arterial stiffness in a person, and apparatus for measuring arterial elasticity and endothelial function in a person; clinical vital sign medical measurement instruments; medical apparatus, namely, fiberoptic light system comprising of an adjustable headlight band with an attached fiber optic cable for providing light from a light source to be used in medical and surgical procedures; medical apparatus, namely, preoperative hair removal clippers and surgical blades for using during surgical procedures; medical devices, namely, needleless intravenous connector for medical fluids; medical devices, namely, molded tubing apparatus for medical use, namely, universal connectors and protective caps, needleless connectors, and intravenous manifold connectors for use in medical fluid control; medical devices that allow intravenous access via a syringe or male luer; medical instruments that monitor the delivery of medication to a patient, that store medication delivery parameters, namely, drug names, drug conflicts, flow rates, dosages, volumes, delivery time periods, patient physiological parameters, vital signs parameters, and that monitor medication delivery parameters selected by a care giver, and that compare the medication delivery parameters selected by a care giver to stored medication delivery parameters, and that provide display data based on the comparison, namely, medication delivery monitors, infusion pumps, syringe pumps, programmable infusion pumps, programmable syringe pumps and the resident control computer programs sold as an integral component thereof; surgical instruments, namely, blade tips, shafts, scissors rasps, and osteotomes; surgical clamps and forceps; thermometers, disposable thermometer covers, thermometer probes, thermometer stands, thermometer probe cover holders, thermometer base units, all for medical purposes; medical device, namely, compression garments; biopsy hypodermic needle for surgical and medical use; medical apparatus, namely, medical fluid conducting apparatus for use with medical infusion pumps; surgical instruments, namely, retractors, graspers and dissectors; medical devices, namely, volumetric pump and controller devices used as part of an administration set for the infusion of medical solutions to patients; surgical instruments; endoscopic telescopes for medical purposes; blood pressure measuring apparatus, namely, disposable blood pressure cuffs; medical instruments for measuring and monitoring blood pressure, pulse and temperature; surgical instruments for cutting, retracting, grasping, holding, occluding, dilating, probing, cannulating, draining, aspirating, suturing and ligating in the various fields of surgical practice, namely, surgical knives, scissors, medical chisels, osteotomes, curettes, dissectors, forcep and periosteal elevators, rongeurs, cutting forceps and punches, medical saws, trocars, biopsy hypodermic needles, medical retractors, both hand held and self-retaining, tenacula and vulsellum forceps, hemostatic artery forceps, ansatomosis clamps, dressing and tissue forceps, needle holding forceps, lacrimal probes for medical purposes, sinus probes, gall duct probes, urethral, ureteral, uterine and mitral valve dilators for medical purposes, suction tubes and cannulas, suture needles and ligating instruments for general surgery, rectal and colon procedures, plastic and reconstructive surgery, thoracic and cardiovascular surgery, gynecological and obstetrical procedures, urologic surgery, microsurgery, ophthalmic surgery, ear and endaural surgery, nasal sinus and reconstruction procedures, oral, tonsil, laryngeal, bronchoscopic and esophagoscopic procedures, pediatric surgery, neurological surgery and orthopaedic surgery; suture hypodermic needles and ligating medical instruments; biopsy hypodermic needles, punches and forceps; anesthesia equipment, namely, medical suction tubes, airways, tube-holding forceps; medical electronic instruments, namely, electronic ocular tonometers, and tonographers; diagnostic doscopes for medical use; surgical supplies, namely, drapes for surgical procedures; surgical equipment, namely, a tray and basin system for disinfecting, transporting and storage of surgical instruments; tourniquet inflation units, namely, medical devices for use in cannulation or to stem the flow of traumatic bleeding, namely, tourniquets; patient examining and treatment tables; surgical supplies, namely, drapes for surgical procedures; surgical equipment, namely, a tray and basin system for disinfecting; medical tubes, vials and envelopes used for transporting fluid bacteriological specimens, for medical purposes, sold empty; medical device for determining coagulation time and parts thereof; medical devices, namely, infusion pumps for delivering measured amounts of solutions into the bloodstream over time; anti-microbial product, namely, antimicrobial needleless access connectors to facilitate the transmission of fluids and antimicrobial IV dressings; and medical devices for obtaining body fluid samples for laboratory use in testing body fluid specimens | Education and training services, namely, classes, seminars and workshops in the fields of medicine, medical research, medical analysis, medical diagnosis, and medical product development; educational services, namely, conducting lectures, workshops, web-based seminars and roundtable discussions focused on key industry trends in healthcare for professionals; training in the operation, repair and maintenance of diagnostic instruments, reagents and software for use with such instruments in the fields of life science research, pharmaceutical development, clinical care and biotechnology; providing online newsletters relating to diabetes healthcare; educational services, namely, providing online instruction in the fields of medical technology, diabetes and diabetes care; educational services, namely, professional training in the field of specimen collection; education services, namely, providing seminars and on-line seminars in the fields of medical technology, safe medical injection and diabetes care, and distributing print and electronic course materials in connection therewith; educational services, namely, providing training in the field of diabetes and diabetes care provided by means of a call center and distribution of printed and electronic training materials in connection therewith; providing education services in the field of women's health and cancer in the form of live and on-line training, classes and seminars; educational services, namely, providing continuing professional education program, namely, lectures, workshops and roundtable discussions focused on medical sample collection, processing and transport, the preanalytical phase of laboratory testing, troubleshooting incorrect handling procedures, and disease etiology; educational services, namely, in-service training programs to educate customers on compliance with the healthcare safety acts designed for healthcare workers who may be exposed to disease while handling certain medical devices, to educate customers on proper use of safety-engineered products, and assist customers with conversions on how to use safety-engineered products; clinical education services, namely, providing mentoring, tutoring, classes, seminars and workshops in the fields of oncology, interventional procedures, malignant pleural effusions, and vertebral augmentation; medical training and teaching

Goods & Services
Drug development support program, namely, pharmaceutical research and development program that provides associated technical and regulatory documents and testing support designed to accelerate time to market of new innovative and biosimilars drugs



Goods & Services
Medical devices and apparatus, namely, syringes, needles, infusion and injection devices for administering drugs, medical scissors, and needle-free injection systems, and replacement parts specially adapted therefor


Goods & Services
Laboratory equipment, namely, sample preparation and real-time polymerase chain reaction (RT-PCR) analyzer for the detection and identification of infectious diseases for clinical diagnostic use for medical purposes

Goods & Services
Laboratory apparatus and instruments, namely, molecular testing apparatus for testing any disease state associated with changes in DNA, RNA or protein content or expression not for medical purposes; molecular diagnostic apparatus for the detection of any disease state associated with changes in DNA, RNA or protein content or expression not for medical purposes; scientific apparatus and instruments, namely, gene library preparation instrument that automates or assists in the preparation of DNA, RNA, and protein for detection; molecular chemistry apparatus and instruments, namely, gene library preparation instrument that automates or assists in the preparation of DNA, RNA, and protein for detection; molecular laboratory apparatus and instruments, namely, library preparation instrument or any device that automates or assists in the preparation of DNA, RNA, and protein for detection; apparatus and instruments for scientific research in molecular laboratories, namely, library preparation instrument or any device that automates or assists in the preparation of DNA, RNA, and protein for detection; apparatus and instruments for use in genotyping for scientific or research use; apparatus and instruments for use in molecular sample preparation for scientific or research use; apparatus and instruments for use in detecting genetic sequencing for scientific or research use; apparatus and instruments for use in compound screening for scientific or research use; apparatus and instruments for use in nucleic acid amplification for scientific or research use; apparatus and instruments for use in gene copy number determination for scientific or research use; apparatus and instruments for use in molecular screening for scientific or research use; apparatus and instruments for use in molecular agricultural applications; apparatus and instruments for use in molecular food security application; apparatus and instruments for use in molecular biological testing not for medical purposes; apparatus and instruments for use in genetic testing not for medical purposes; apparatus and instruments for use in molecular biology not for medical purposes; apparatus and instruments for use in molecular biological processing not for medical purposes; apparatus and instruments for use in molecular biological analysis not for medical purposes; structural parts and fittings for the aforesaid goods | Medical apparatus and instruments, namely, devices for molecular preparation for medical purposes; molecular instruments used to test, measure and detect any disease state associated with changes in DNA, RNA or protein content or expression for medical use; molecular diagnostic apparatus for the detection of DNA, RNA, and protein for medical purposes; medical molecular analysis instruments for the analysis of disease markers that result in changes to DNA, RNA or protein content or sequences; molecular testing instruments for testing changes in DNA, RNA or protein content or sequences for medical purposes; molecular screening instruments for detecting changes in DNA, RNA or protein content or sequences for medical purposes; molecular preparation testing instruments for detecting changes in DNA, RNA or protein content or sequences for medical purposes; structural parts and fittings for the aforesaid goods

Goods & Services
wound dressing, namely, pledgets; urine collection kit comprised of medical diagnostic test strips; medical adhesive tape to secure dressings; medical dressings in the nature of iv dressings; biological tissue grafts; surgical mesh composed primarily of living tissue, namely, soft tissue reconstruction mesh, plug and patches; hemostatics for medical purposes; hemostatics for medical purposes, namely, absorbable hemostats particles for use as a surgical wound dressing; hemostat flour, namely, absorbable collagen hemostat for use as a surgical wound dressing; hemostat sheets, namely, absorbable collagen hemostat for use as a surgical wound dressing; hemostatics for medical purposes, namely, collagen sponges; biomaterial surgical sealants; pre-loaded hemostatic applicators for the application of hemostatic powder
Goods & Services
Promoting public awareness of the overuse and misuse of antibiotics


Goods & Services
Computer software and hardware used in the management of medications, medical devices, and supplies in hospitals and other healthcare institutions; Business consulting and business data analysis services featuring the management of medications, medical devices, and supplies in hospitals and other healthcare institutions for business or commercial purposes

Goods & Services
Computer software and hardware used in the management of medications, medical devices, and supplies in hospitals and other healthcare institutions; Business consulting and business data analysis services featuring the management of medications, medical devices, and supplies in hospitals and other healthcare institutions for business or commercial purposes

Goods & Services
Laboratory instrument and apparatus, namely, single-cell gene expression platform used to capture and analyze individual cells; Reagents for use with flow cytometers for scientific and research use

Goods & Services
drug delivery systems, namely, patch injectors, patch pumps and microinfusors

Goods & Services
Laboratory instrument, namely, automated sample preparation systems comprised of flow cytometers and flow-based analyzers, robotic plate and tube grippers, fluid handling systems comprised of pumps and valves to provide fluid for the cell wash carousel and pipettor wash stations, and comprehensive software interface for providing cell and particle analysis, detection, or counting for scientific, laboratory, and general research uses | Medical instrument, namely, automated sample preparation system comprised of flow cytometers and flow-based analyzers, automated pipettors, fluid handling systems comprised of pumps and valves to provide fluid for the cell wash carousel and pipettor wash stations, robotic plate and tube grippers with integrated software for providing cell and particle analysis, detection, or counting for medical clinical use