
DIASORIN S.P.A.
15 Marks
First Filed:Oct 15, 1998Latest Filed:May 23, 2025Address:VIA CRESCENTINO SNC, SALUGGIA, 13040, IT
Portfolio Overview
Registered
10(67%)Pending
1(7%)Dead
4(27%)Top Classes
Class 10
Medical ApparatusClass 5
PharmaceuticalsClass 1
Industrial Chemicals & AdhesivesTTAB Proceedings
No litigation activity
Top Firms
Haug Partners, LLP(Aug 2023)
6
The Sladkus Law Group(Nov 2021)
6
Ladas & Parry LLP(Mar 2009)
1
ABBOTT LABORATORIES(Nov 2004)
1
Trademark Activity Timeline(1998 – 2025)
- Filings
- Registrations
Trademark Portfolio
15 results
(current & previous marks)
Refine by Mark/Description🔽
Filed
Registered
Last Activity
Filters
Ownership
15
0
Status
10
1
3
1
0
Mark Type
11
2
0
2
0
9
9
8
9
1

Goods & Services
Class 009: Software applications for medical diagnostics; computer software for medical diagnostics; data-processing software; apparatus, equipment, hardware and computers for diagnostics; data storage, processing and transmission apparatus for diagnostic purpose; readers [data processing equipment] for diagnostic purpose; research laboratory analyzers for measuring, testing and analyzing blood and other bodily fluids; luminescence analyzers; apparatus and instruments for scientific research in laboratories; laboratory instruments for the detection of pathogens and toxins in a biological sample for research use; calibrating apparatus; test tubes; holders for test tubes; parts and fittings for the aforementioned goods, included in this class, all applied to the medical diagnostic field.; Class 010: Testing apparatus for medical purposes; diagnostic apparatus for medical purposes; medical devices for diagnostics; medical testing equipment; apparatus for use in medical analysis; electronic analyzers for medical purposes; diagnostic apparatus for the identification of virus, bacterial infections and infectious diseases; laboratory apparatus for medical diagnostic purposes; medical diagnostic apparatus for testing for viruses; diagnostic apparatus for the identification of sexually transmitted infections; automatic analyzers for medical diagnosis; immunological analysers; analysers for bacterial identification for medical purposes; apparatus for analysing bacteria in biological samples [for medical use]; diagnostic equipment for the identification and treatment of hepatitis and retroviruses, metabolic disorders, gastrointestinal diseases, infectious diseases, tumour markers and endocrine disorders.

Owner:
Serial:79384704
Filed:Aug 22, 2023
Classes:1, 5, 9, +2
Registration:7546937
Registered:Oct 29, 2024
Goods & Services
Semiconductor testing apparatus; Laboratory apparatus, namely, analyzers for measuring and analyzing blood and other bodily fluids; Scientific research specimen collection devices in the nature of tubes, swabs and bags for the collection of blood, urine and fecal matter; Data processing equipment, namely, optical readers for diagnostic purposes other than for medical use; Measuring or testing machines and instruments for diagnostic purposes, other than for medical use, namely, apparatus composed of PC tablets and optical readers for testing liquids and solids; Downloadable portable and cloud-connected point of care (POC) immunodiagnostic software platforms for diagnostic research and scientific use; Computer software and hardware for the design, creation and testing of medical diagnostic instruments; Downloadable software for processing data; diagnostic apparatus for the detection of cancer; computer hardware and computers; Downloadable computer software using artificial intelligence for use in software development in the field of healthcare; Downloadable computer software for medical diagnostics; Tablet computers for testing and diagnostic purposes; Diagnostic apparatus for the detection of pathogens, not for medical purposes; Disposable dispenser syringes for laboratory use; Downloadable computer software applications for diagnosing illnesses; Scientific apparatus and instruments other than for medical purposes for immunological analysis and diagnostics; Downloadable computer software for diagnosing illnesses; Computer software platforms, downloadable, for diagnosing illnesses; Computer software platforms, recorded, for diagnosing illnesses; Downloadable computer programs for diagnosing illnesses in the diagnostic field; Laboratory robots; Pathogen measurement and blood analysis machines and equipment for use in diagnosing illnesses; Downloadable software for processing and interpreting data in the field of medical and veterinary diagnostics; Chemical reagents for medical or veterinary purposes; Diagnostic biomarker reagents for medical purposes; Diagnostic agents for pharmaceutical use; Diagnostic agents for medical, clinical and pharmaceutical purposes; Diagnostic agents for medical use; Diagnostic test kits consisting primarily of medical diagnostic reagents for detecting antibodies, enzymes, antigens, diseases, viruses and infections, for medical and clinical purposes; Diagnostic reagents for serologic tests of infections for medical and clinical purposes; Diagnostic kits comprised of bioreagents for use in disease testing; Diagnostic test kits comprised of reagents and assays for testing body fluids; In vitro diagnostic test kit comprised of medical diagnostic reagents for testing of body fluids; Diagnostic testing materials for medical use in the form of reagent paper for measuring antibodies and blood cells; Biochemical preparations for medical or veterinary purposes for treating respiratory syndromes; Bacterial preparations for medical or veterinary use; Reagents and medical diagnostic test strips for detecting nucleic acid targets and indicators of disease, namely, enzymes, antigens, antibodies and nucleic acids; Medical diagnostic reagents and assays for testing of body fluids; Immunoassay reagents for medical use; Diagnostic preparations for in vitro testing for medical use; Medical diagnostics testing for the detection, diagnosis, analysis, and confirmation of viruses and infections; Medical diagnostic testing in antibody detection; Reagent paper for medical purposes; Cultures of microorganisms for medical or veterinary use; Preparations of microorganisms for medical or veterinary purposes; Bacteriological preparations for medical or veterinary use; Diagnostic preparations for medical purposes; Diagnostic reagents for medical use; Biological and chemical diagnostic reagents for medical or veterinary purposes; Diagnostic reagents for medical or veterinary purposes; Diagnostic reagents for medical purposes; Diagnostic preparations for medical or veterinary purposes; In vitro diagnostic preparations for medical use; Swabs and diagnostic preparations for medical and medical diagnostic use; Nasopharyngeal swabs for medical use; Chemico-pharmaceutical preparations for medical diagnostic use; Pharmaceuticals for use in the diagnosis of infectious diseases; Pharmaceutical, medical diagnostic and veterinary preparations for use in the diagnosis of infectious diseases; Disinfectants; Dietetic substances being food and beverages adapted for medical use; Chemicals used in science; Diagnostic biomarkers for use in the assessment of infectious diseases for analytical, research and scientific use; Diagnostic kits comprised of specimen receptors and medical diagnostic reagents for testing for selected bacteria; Diagnostic testing materials, namely, preparations and reagents for scientific use and research use other than for medical purposes; Chemical reagent mixtures, other than for medical or veterinary use; Diagnostic preparations for use in assessment of toxicological property of chemical compounds and mixture, for scientific use; Diagnostic chemicals for use in antibiotic susceptibility testing for scientific research use, other than for medical purposes; Diagnostic preparations for research laboratory use; Diagnostic preparations for in vitro testing, for scientific use; Chemical reagents for molecular biology, other than for medical or veterinary purposes; Chemical reagents for use in analytical tests, other than for medical or veterinary purposes; Chemical reagents in kit form for conducting immunosorbent assays for scientific purposes; Chemical reagents, namely, solutions used in diagnostic test kits for laboratory or research purposes; Diagnostic substances for scientific or research use, other than for medical purposes; Reagent bearing diagnostic test strips for testing blood samples and body fluids; Chemically treated non-medical test strips for laboratory or research purposes; Reagent paper, other than for medical or veterinary purposes; Cultures of microorganisms, other than for medical or veterinary use; Preparations of microorganisms, other than for medical or veterinary use; Bacterial preparations, other than for medical or veterinary use; Bacteriological preparations, other than for medical or veterinary use; Biological preparations, other than for medical or veterinary purposes; Chemical substances for analyses in laboratories, other than for medical or veterinary purposes; Chemical reagents, other than for medical or veterinary purposes; Reagents for chemical analyses; Diagnostic reagents for scientific use; Biochemical reagents used for non-medical purposes; Diagnostic reagents, other than for medical laboratory use; Diagnostic reagents and preparations not for medical or veterinary purposes; Reagents for use in scientific apparatus for chemical or biological analysis; Cell culture reagents for scientific and research use; Surgical apparatus and instruments for medical, pharmaceutical and veterinary diagnostic use; Diagnostic apparatus for the identification of infectious diseases; Diagnostic imaging apparatus for medical use; Diagnostic apparatus for identifying antibodies against the SARS-CoV-2 virus in capillary blood samples; Medical diagnostic apparatus for testing antigens for COVID-19 diagnosis; Medical diagnostic apparatus for testing molecular cells for COVID-19 diagnosis; Medical diagnostic apparatus for testing DNA and RNA samples for medical purposes; Medical diagnostic instruments for the analysis of body fluid samples; Blood testing apparatus, namely, blood sampling tubes; Genetic testing apparatus for medical purposes; Diagnostic apparatus for medical purposes used in medical laboratories for testing DNA; Point of care (POC) immunochemical testing apparatus for carrying-out diagnostic tests for medical purposes; Laboratory apparatus for medical diagnostic purposes in the nature of liquid- and cell- dispensing devices for analyzing and investigating cells; Medical devices for medical diagnostic, examination and monitoring in clinical application sectors, namely, point of care diagnostic, examination and monitoring devices for ascertaining clinically relevant measurement of analyte concentration in whole blood and blood plasma; Diagnostic medical apparatus for testing samples of DNA and RNA; Saliva collection devices for medical diagnostic purposes in the nature of saliva vials; Extraction syringes for medical purposes; Injection syringes; Analysers for bacterial identification for medical purposes; Blood testing apparatus; Medical diagnostic apparatus for testing for viruses; Updating of computer software in the medical diagnostic field; Design and development of computer software in the medical diagnostic field; Rental of computer software for medical diagnostic purposes; Maintenance and repair of computer software in the medical diagnostic field; Design and development of computer hardware in the medical diagnostic field; Design and development of software and hardware in the medical diagnostic field; Scientific research and development in the field of biotechnology; Research and development in the field of medical diagnostic preparations; Biological research; Chemical research; Scientific research; Computer aided medical diagnostic testing services; Scientific laboratory services; Chemical analysis; Installation of computer software for use in the medical diagnostic field; Bacteriological research; Stem cell research services and laboratory testing of materials

Owner:
Serial:79383235
Filed:Jul 28, 2023
Classes:1, 5, 9, +1
Registration:7525844
Registered:Oct 8, 2024
Goods & Services
Downloadable computer software mobile applications for use in diagnosing individuals; Downloadable computer software for medical diagnostics and for operating diagnostic apparatus that is not for medical purposes; Testing machines and instruments for diagnostic purposes, other than for medical use, namely apparatuses composed of PC tablets and optical readers for testing liquids and solids; Downloadable portable and cloud-connected point of care (POC) immunodiagnostic software platforms for diagnosing individuals; Downloadable computer software and hardware for the design, creation and control of tools for medical diagnostics; Downloadable software for processing data; Apparatus, equipment, hardware and computers for diagnostics; Data storage, processing and transmission apparatus for diagnostic purposes, namely, blank tapes for storing computer data; Data processing equipment, namely, optical readers for diagnostic purposes; PC tablets used for test and diagnostic purposes; Video monitors; Testing apparatus not for medical purposes, namely, apparatus for testing liquids and solids; Measuring or testing machines and instruments for diagnostic purpose, other than for medical use, namely, apparatus composed of PC tablets and optical readers for testing liquids and solids; Research laboratory analyzers for measuring, testing and analyzing blood and other bodily fluids; Medical diagnostic test reagents for testing body fluids for veterinary use; Medical diagnostic test reagents for testing body fluids for medical purposes; Diagnostic agents for medical use; Chemical reagents for medical or veterinary purposes; Diagnostic kits comprising medical diagnostic bioagents for extracting, testing and analyzing body fluids; Diagnostic test kits comprising medical diagnostic reagents and assays for testing of body fluids for use in disease detection, namely, infectious disease, and detection of biological parameters; Diagnostic testing materials for medical use, namely, medical diagnostic test strips for use in the field of infectious diseases and detection of biological parameters; Medical diagnostic test strips for blood analysis; Virus testing preparations for medical use; Medical diagnostic assays for medical use; In vitro medical diagnostic tests for use in disease testing; In vitro medical diagnostic assays for use in disease testing; In vitro medical diagnostic test kits consisting primarily of monoclonal antibodies, buffers and reagents for use in disease testing; In vitro medical diagnostic test strips for blood analysis; In vitro medical diagnostic reagents; Medical diagnostic test strips for the detection, diagnosis, screening and confirmation of viruses and infections; Reagents and test strips for medical purposes for use as indicators of disease and such indicators being antigens and antibodies; Immunoassay reagents for medical use; Biological and chemical preparations and reagents for medical or veterinary purposes for the detection of infectious diseases; Biochemical preparations for medical or veterinary purposes for the detection of infectious diseases; Bacterial preparations for medical and veterinary use; Nucleic acid sequences for medical and veterinary purposes; Medical diagnostic regents for the detection of pathogens responsible for respiratory infections in the nature of influenza, COVID-19 and Group A streptococcus; Medical diagnostic reagents for the detection of pathogens responsible for sexually transmitted infections; Chemicals used in science; Chemical diagnostic reagents for scientific purposes; Diagnostic preparations, other than for medical or veterinary purposes; Reagent bearing diagnostic strips for testing of infectious diseases and organic molecules, other than for medical or veterinary use; Diagnostic testing materials, other than for medical use in the nature of reagent bearing diagnostic strips for testing saliva; Control preparations, namely, chemical control solutions for in vitro diagnostic testing, screening, confirmation and analysis for laboratory or research use; Diagnostic reagents and chemical reagents for scientific or research purposes; Solutions in the nature of chemical control solutions used in diagnostic test kits for laboratory or research purposes; Chemically treated non-medical test strips for laboratory or research purposes; Diagnostic preparations for laboratory or research use; Diagnostic preparations for use in assessment of toxicological property of chemical compounds and mixture for scientific use; Diagnostic biomarkers for analytical, research and scientific use; Diagnostic preparations for in vitro testing for scientific use; nucleic acids; nucleic acids for scientific purposes; Diagnostic testing apparatus for medical purposes for testing biomarkers and biomolecules; Medical diagnostic apparatus for medical purposes; Tools for medical diagnostics, namely, medical diagnostic apparatus for testing for viruses and infectious disease; Medical devices for diagnosing viruses; Medical testing equipment, namely, medical device for detecting antibodies against SARS-CoV-2; Medical apparatus for medical analysis of bacteria; Electronic analyzers for bacterial identification for medical purposes; Apparatus for taking blood samples; Apparatus for taking body fluid samples; Point of care (POC) diagnostic, examination and monitoring equipment, namely, respiratory equipment and monitors therefor; Point of care (POC) immunochemical testing apparatus for carrying out diagnostic tests for medical purposes; Diagnostic apparatus for the identification of specific antibodies against SARS-CoV-2 in capillary blood samples; Diagnostic apparatus for the identification of virus, bacterial infections and infectious diseases; Radiological diagnostic imaging apparatus for medical purposes in the field of immunochemistry; Medical diagnostic apparatus for testing for viruses; Apparatus for DNA and RNA testing for medical purposes; Syringes for injections; Extraction medical syringes; Disposable hypodermic syringes for medical use; Medical syringe needles; Medical diagnostic apparatus for the identification of respiratory infections in the nature of influenza, SARS-CoV-2 and Group A streptococcus; Medical diagnostic apparatus for the identification of sexually transmitted infections; Molecular platform designed for the detection of pathogens causing infectious diseases through PCR nucleic acid amplification technology
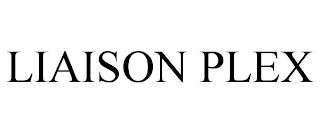
Owner:
Serial:98062754
Filed:Jun 28, 2023
Classes:1, 5, 9, +1
Registration:7972029
Registered:Oct 7, 2025
Goods & Services
Downloadable computer software mobile applications for use in diagnosing individuals; Downloadable computer software for medical diagnostics and for operating diagnostic apparatus that is not for medical purpose; Testing machines and instruments for diagnostic purposes, other than for medical use, namely, apparatuses composed of PC tablets and optical readers for testing liquids and solids; Downloadable portable and cloud-connected POC (Point of care) immunodiagnostic software platform for diagnostic research and scientific use; Downloadable computer software and hardware for the design, creation and control of tools for medical diagnostics; Downloadable software for data processing; data processing equipment, namely, optical readers for diagnostic purposes; PC tablets used for test and diagnostic purpose; Video monitors; Testing apparatus not for medical purposes, namely, apparatus for testing liquids and solids; measuring or testing machines and instruments for diagnostic purpose, other than for medical use, namely, apparatus composed of PC tablets and optical readers for testing liquids and solids; research laboratory analyzers for measuring, testing and analyzing blood and other bodily fluids; Medical diagnostic preparations for medical and veterinary purposes for the detection of infectious diseases; Medical diagnostic test reagents for testing body fluids for veterinary use; Medical diagnostic test reagents for testing body fluids for medical purposes; Diagnostic agents for medical use; Chemical reagents for medical or veterinary purposes; Diagnostic kits comprising medical diagnostic bioreagents for extracting, testing and analyzing body fluids; diagnostic test kits comprising medical diagnostic reagents and assays for testing of body fluids for use in disease detection, namely, infectious disease, and detection of biological parameters; Diagnostic testing materials for medical use, namely, medical diagnostic test strips for use in the field of infectious diseases and detection of biological parameters; Medical diagnostic test strips for blood analysis; Virus testing preparations for medical use; Medical diagnostic assays for medical use; In vitro medical diagnostic tests for use in disease testing; In vitro medical diagnostic assays for use in disease testing; In vitro medical diagnostic test kits consisting primarily of monoclonal antibodies, buffers and reagents for use in disease testing; in vitro medical diagnostic test strips for blood analysis; in vitro medical diagnostic reagents; medical diagnostics tests strips for the detection, diagnosis, screening, and confirmation of viruses and infections; reagents and test strips for medical purposes for use in detecting the presence of nucleic acid targets, and as indicators of diseases with such indicators being enzymes, antigens, antibodies and nucleic acids; immunoassay reagents for medical use; Biological and chemical preparations and reagents for medical or veterinary purposes for the detection of infectious diseases; biochemical preparations for medical or veterinary purposes for the detection of infectious diseases; bacterial preparations for medical and veterinary use; Nucleic acid sequences for medical and veterinary purposes; Test cartridges in the nature of medical diagnostic reagents for the detection of pathogens responsible for respiratory and gastrointestinal infection syndromes and for bacterial and fungal sepsis; Chemicals used in science; chemical diagnostic reagents for scientific purposes; Diagnostic preparations, other than for medical or veterinary purposes; Reagent bearing diagnostic strips for testing of infectious diseases and organic molecules, other than for medical or veterinary use; diagnostic testing materials, other than for medical use in the nature of reagent bearing diagnostic strips for testing saliva; Control preparations, namely, chemical control solutions for in vitro diagnostic testing, screening, confirmation and analysis for laboratory or research use; diagnostic reagents and chemical reagents for scientific or research purposes; solutions in the nature of chemical control solutions used in diagnostic test kits for laboratory or research purposes; Chemically treated non-medical test strips for laboratory or research purposes; diagnostic preparations for laboratory or research use; diagnostic preparations for use in assessment of toxicological property of chemical compounds and mixture for scientific use; diagnostic biomarkers for analytical, research and scientific use; diagnostic preparations for in vitro testing for scientific use; Nucleic acids; Nucleic acids for scientific purposes; Diagnostic testing apparatus for medical purposes for testing biomarkers and biomolecules; Medical diagnostic apparatus for medical purposes; tools for medical diagnostics, namely, medical diagnostic apparatus for testing viruses and infectious disease; medical devices for diagnosing viruses; Electronic analyzers for bacterial identification for medical purposes; apparatus for taking blood samples; apparatus for taking body fluid samples; Point of Care (POC) diagnostic, examination, and monitoring equipment, namely, respiratory equipment and monitors therefor; Point of care (POC) immunochemical testing apparatus for carrying-out diagnostic tests for medical purposes; diagnostic apparatus for the identification of virus, bacterial infections and infectious diseases; Radiological diagnostic imaging apparatus for medical purposes in the field of immunochemistry; laboratory apparatus for medical diagnostic purposes in the nature of cell analyzers used for the purpose of diagnosing viruses; medical diagnostic apparatus for testing for viruses; apparatus for DNA and RNA testing for medical purposes; Syringes for injections; hypodermic syringes for medical use; extraction medical syringes; disposable hypodermic syringes for medical use; medical syringe needles; Syringe needles for medical purposes; micro-needling needles for medical purposes; Medical diagnostic apparatus for the identification of pathogens responsible for syndromes of respiratory and gastrointestinal infections and for bacterial and fungal sepsis; Molecular platform designed for the detection of pathogens through multiplex PCR nucleic acid amplification technology; X-ray diagnostic apparatuses

Goods & Services
"DETECT"
Goods & Services
Chemicals for use in science; chemical diagnostic reagents for scientific purposes; Diagnostic preparations, other than for medical or veterinary purposes; diagnostic substances, other than for medical or veterinary use; Reagent bearing diagnostic strips for testing of infectious diseases and organic molecules, other than for medical or veterinary use; diagnostic testing materials, other than for medical use in the nature of reagent bearing diagnostic strips for testing saliva; Control preparations, namely, chemical control solutions used in in-vitro diagnostic testing, screening, confirmation and analysis for laboratory or research use; diagnostic reagents and chemical reagents for laboratory or research purposes; chemical control solutions used in diagnostic test kits for laboratory or research purposes; chemically treated non-medical test strips for laboratory or research purposes; diagnostic preparations for laboratory or research use; diagnostic preparations for use in assessment of toxicological property of chemical compounds and mixture for scientific use; diagnostic biomarkers for analytical, research and scientific use; diagnostic preparations for in vitro testing for scientific use | Diagnostic preparations for medical and veterinary purposes; Medical diagnostic test reagents for testing body fluids for veterinary use; medical diagnostic test reagents for testing body fluids for medical purposes; Diagnostic agents for medical use; Chemical reagents for medical or veterinary purposes; diagnostic kits comprising medical diagnostic bioreagents for extracting, testing and analyzing body fluids; diagnostic test kits comprising medical diagnostic reagents and assays for testing of bodily fluids for use in diseases detection, namely, infectious diseases and detection of biological parameters; Diagnostic testing materials for medical use, namely, medical diagnostic test strips for use in the field of infectious deseases and detection of biological parameters; medical diagnostic test strips for blood analysis; virus testing preparations for medical use; medical diagnostic assays for medical use; in vitro medical diagnostic tests, namely, test strips; in vitro medical diagnostic assays for medical use; in vitro medical diagnostic test kits consisting primarily of monoclonal antibodies, buffers, and reagents for use in disease testing; in vitro medical diagnostic test strips for blood analysis; in vitro medical diagnostic reagents; medical diagnostics tests strips for the detection, diagnosis, screening, and confirmation of viruses and infections; reagents and test strips for medical purposes for use in detecting the presence of nucleic acid targets, and indicators of diseases, such indicators including enzymes, antigens, antibodies and nucleic acids; immunoassay reagents for medical use; biochemical preparations for medical or veterinary purposes for detection of infectious diseases and biological parameters; bacterial preparations for medical and veterinary use | Downloadable computer software mobile applications for use in diagnosing individuals; downloadable computer software for medical diagnostics and for use to operate diagnostic apparatuses not for medical purpose; downloadable portable and cloud-connected POC (Point of care) immunodiagnostic software platform for diagnosing individuals; Downloadable computer software and hardware for the design, creation and control of tools for medical diagnostics, data-processing software, apparatus, equipment, hardware and computers for diagnostics; data storage, processing and transmission apparatus for diagnostic purpose, namely, blank tapes for storage of computer data; electronic book readers for diagnostic purpose; PC tablets used for test and diagnostic purpose; Video monitors; structural parts and fittings for the aforementioned goods where applicable, all applied in the diagnostic field; Testing apparatus not for medical purposes, namely, apparatus for testing liquids and solids; testing machines and instruments for diagnostic purpose, other than for medical use, namely, apparatus for testing liquids and solids, composed of PC tablets and optical reader; research laboratory analyzers for measuring, testing and analyzing blood and other bodily fluids | Diagnostic testing apparatus for medical purposes for testing biomarkers and other biomolecules; medical diagnostic apparatus for medical purposes; tools for medical diagnostics, namely, medical diagnostic apparatus for testing for viruses and infection diseases; medical devices for diagnosing viruses; medical testing equipment, namely, medical device for detecting antibodies against SARS-CoV-2; medical apparatus for use in medical analysis for analyzing bacteria; Electronic analyzers for bacterial identification for medical purposes; apparatus for taking blood samples; apparatus for taking body fluid samples; Point of Care (POC) diagnostic, examination, and monitoring equipment, namely, respiratory equipment and monitors therefor; Point of care (POC) immunochemical testing apparatus for carrying-out diagnostic tests for medical purposes; diagnostic apparatus for the identification of specific antibodies against SARS-CoV-2 in capillary blood samples; medical diagnostic apparatus for the identification of antigens to diagnose Covid-19 via nasal swab; diagnostic apparatus for the identification of virus and infectious diseases; radiological diagnostic imaging apparatus for medical purposes in the field of immunochemistry; medical diagnostic apparatus for testing for viruses; apparatus for DNA and RNA testing for medical purposes; Syringes for injections; syringes for medical purposes; hypodermic syringes; extraction medical syringes; disposable hypodermic syringes for medical use; medical syringe needles; needles for medical purposes; medical micro-needling devices
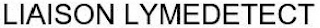
Goods & Services
"LYME DETECT"

Goods & Services
Scientific apparatus and instruments, namely, molecular biological [ and immunological ] testing systems, namely, [ microarray scanners, ] DNA [ and protein chips, ] kits for multiplex polymerase chain reaction (PCR) tests consisting primarily of optical and electronic apparatus for analyzing biological or biochemical samples, namely, [ electrophoresis apparatus, ] nucleic acid and [ protein sequencing apparatus, ] and polymerase chain reaction (PCR) thermal cyclers, including associated hardware and software therefor and also containing devices for removing, marking, applying and identifying biological or biochemical samples, pipettes, [ or cannulae; ] diagnostic testing apparatus for scientific and research testing of biological or biochemical samples, namely, electrophoresis apparatus, nucleic acid [ and protein sequencing apparatus,] and polymerase chain reaction (PCR) thermal cyclers; computer systems and software for collecting, storing, analysing and reporting biological information, and for sample tracking and managing projects, laboratory workflows and data, for use in the fields of scientific, diagnostic and clinical research and for clinical diagnostic purposes; software for electronic medical and diagnostic apparatus; software, namely, databanks and computer programs containing algorithms for helping to make medical decisions or carry out research, for diagnostic and therapeutic applications; Computer hardware for medical and diagnostic tasks; devices for generating images for detecting, quantifying and linking biological or chemical molecules; laboratory instruments, [ namely, genome sequencers; ] devices for generating images, namely, [ fluorimeters used to register ] fluorescence signals and produce data; [ Diagnostic preparations for medical purposes; diagnostic materials in the nature of medical diagnostic reagents and assays for extraction, testing and analysis of bodily fluids, namely, extraction of viral nucleic acid, bacterial nucleic acid, stool DNA, blood DNA, and RNA, and for the extraction and isolation of blood cells; preparations for use in medical diagnostic tests; chemical preparations for testing blood for medical purposes; chemical preparations for testing blood for veterinary purposes; in vitro diagnostic preparations for medical purposes; in vitro diagnostic preparations for veterinary purposes; chemicals for medical use for testing DNA; nucleic acid sequences for medical and veterinary purposes; reagents for use in diagnostic tests for medical purposes; reagents for use in diagnostic tests for veterinary purposes; reactants for medical diagnosis; diagnostic reagents for medical use; diagnostic biomarker reagents for medical purposes; diagnostic biomarker reagents for veterinary purposes; diagnostic substances for medical use; diagnostic substances for veterinary use; antiserums for diagnostic purposes; chemical test reagents for medical use; chemical test reagents for veterinary use; reagents for microbiological analysis for medical purposes; reagents for microbiological analysis for veterinary purposes; reagents for use with analyzers for medical purposes; reagents for use with analyzers for veterinary purposes; diagnostic chemical reagents for medical use; diagnostic chemical reagents for veterinary use; medical diagnostic reagents and assays for testing of body fluids; diagnostic agents for pharmaceutical use; test strips for measuring blood glucose levels; medical diagnostic test strips; chemical diagnostic reagents for medical use for extracting, testing and analyzing body fluids, nucleic acids, DNA, RNA; chemical diagnostic reagents for medical use for extracting, isolating, testing and analyzing body fluids ]; "MDX"; [ Test kits for clinical medical use comprising test tubes, test plates and indicator diagrams for evaluating test results; apparatus for carrying out medical diagnoses, namely, fluorescence diagnostic apparatus and instruments used for the purpose of identifying infectious diseases and the severity thereof; diagnostic imaging apparatus for medical use; diagnostic lasers for medical use; medical and diagnostic devices, apparatus and equipment for molecular analysis; scanners for medical diagnosis; spectrum analyzers for use in medical diagnosis; Ultrasonic medical diagnostic apparatus being sources of ultrasonic fields for use in medical diagnosis; Magnetic apparatus being sources of magnetic fields for use in medical diagnosis; Electromagnetic medical diagnostic imaging apparatus being sources of electromagnetic fields for use in medical diagnosis; optical instruments for medical diagnostics; containers for disposal of medical instruments, namely, medical samples, syringes and plungers; Medical sampling vials of plastic or other materials for containing medical samples; Medical preparation vials of plastic or other materials for preparation of medical samples; diagnostic testing instruments for medical purposes, namely, electrical laboratory diagnostic instruments for the analysis of nucleic acids and proteins for medical use; diagnostic kits consisting primarily of electronic instruments for medical diagnosis and analysis of body fluids, tissues, biomolecules, DNA, antibodies, proteins, molecular material or cellular material; scientific apparatus for diagnostic testing of biological samples medical use; medical specimen cups ]
Owner:
Serial:87399567
Filed:Apr 5, 2017
Classes:1, 5, 9, +1
Registration:5868840
Registered:Sep 24, 2019
Goods & Services
(Based on 44(e)) Scientific apparatus, namely, molecular biological and immunological testing systems, namely, microarray scanners, DNA and protein chips, kits for multiplex polymerase chain reaction (PCR) tests consisting primarily of optical and electronic apparatus for analyzing biological or biochemical samples, namely, electrophoresis apparatus, nucleic acid and protein sequencing apparatus, and polymerase chain reaction (PCR) thermal cyclers, including associated hardware and software therefor and also containing devices for removing, marking, applying and identifying biological or biochemical samples, pipettes, or cannulae; (Based on 1(a)) Diagnostic kits comprised of medical diagnostic reagents and assays for extraction, testing and analysis of bodily fluids, namely, extraction of viral nucleic acid, bacterial nucleic acid, stool DNA, blood DNA, and RNA, and for the extraction and isolation of blood cells; medical diagnostic reagents; diagnostic preparations for medical purposes; nucleic acid sequences for medical and veterinary purposes; diagnostic preparations for veterinary purposes; diagnostic preparations for medical and veterinary purposes; (Based on 1(a)) Diagnostic reagents for clinical or medical laboratory use; (Based on 44(e)) Medical diagnostic instruments for the analysis of body fluids, tissue, biomolecules, DNA, antibodies, proteins, molecular material, or cellular material; Medical apparatus and instrument for diagnostic use, namely, apparatus for medical diagnostic testing in the fields of body fluid diagnostic testing, tissue-based diagnostic testing, molecular testing, protein testing, antibody testing, cytology and cell-based testing; electrical laboratory diagnostic instruments and apparatus for the analysis of nucleic acids and proteins for medical use; diagnostic kits consisting primarily of electronic instruments for medical diagnosis and analysis of body fluids, tissues, biomolecules, DNA, antibodies, proteins, molecular material or cellular material

Goods & Services
Pharmaceutical products and substances all for diagnostic testing in the fields of medicine [ or veterinary science ]; diagnostic reagents for medical use, diagnostic reagents in kit form for medical use, chemical reagents used for medical diagnosis and analysis; in vitro diagnostic kits consisting primarily of consumable, buffers and reagents; In vitro diagnostic kits consisting primarily of consumable, buffers and reagents; surgical, medical, [ dental and veterinary ] apparatus and instruments, all for diagnostic testing; diagnostic instruments for medical analysis
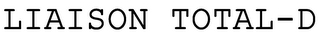
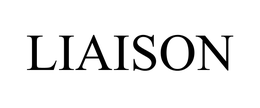
Owner:
Serial:78066444
Filed:May 30, 2001
Classes:1, 5, 9, +1
Registration:2978132
Registered:Jul 26, 2005
Goods & Services
Apparatus and equipment for diagnostic research in the scientific field, namely, mixers, washing apparatus incorporating a suction device, gamma scintilation counter, gamma counter, test tubes and racks for tubes, horizontal shaker for the test tube racks, micro pipettes; computers and computer programs for diagnostic research, namely for controlling diagnostic processes, displaying, transmitting, storing, evaluating, analyzing, interpreting and processing of diagnostic data and for creating quality control reports; [ Diagnostic reagents for pharmaceutical and veterinary use ]; Diagnostic reagents for scientific or research use; Instruments and apparatus for diagnostic examination in the medical field, namely washing devices, plastic tweezers, text instruments for immunoassays, mixers, suture materials

Goods & Services
Medical diagnostic reagents and raw materials for in vitro use in testing laboratories; Diagnostic preparations and reagents for research and scientific use; reagents and raw materials used in research and development of in vitro medical diagnostic apparatus; in vitro medical diagnostic apparatus

