
EVOLVE BIOLOGICS INC.
5 Marks
Corp.
First Filed:Jul 25, 2013Latest Filed:Dec 12, 2017Address:Mississauga, Ontario, L5N 8G6, CA
Portfolio Overview
Registered
5(100%)Top Classes
Class 5
PharmaceuticalsClass 41
Education and EntertainmentClass 42
Software, IT Services & Scientific ResearchTTAB Proceedings
No litigation activity
Top Firms
Merchant & Gould P.C.(Dec 2017)
5
Trademark Portfolio
5 results
(current & previous marks)
Refine by Mark/Description🔽
Filed
Registered
Last Activity
Filters
Ownership
5
0
Status
5
0
0
0
0
Mark Type
3
1
0
1
0
3
5
3
5
5

Owner:
Serial:87717949
Filed:Dec 12, 2017
Classes:5, 37, 40, +2
Registration:6297748
Registered:Mar 23, 2021
Goods & Services
Class 005: Biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for human clinical use, namely, plasma protein fractions, serum albumin, immunoglobulins, coagulation factors, antihemophilic factors, clotting factors, enzymes, coenzymes, coactivators, inhibitors, and structural proteins; resins, for medical use, namely, polysaccharide beads cross-linked to ligands for capturing proteins from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate using chromatographic methods; Class 037: Construction of biotech, biopharmaceutical and plasma manufacturing facilities relevant to the manufacturing of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate; consulting services concerning construction of biotech; Consulting services concerning building construction of biotech facilities; Class 040: Contract manufacturing services for others for biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, and clinical use; consulting services concerning manufacturing of biologics; Class 041: Educational services, namely, designing, arranging, and conducting training programs for research and development, manufacturing of biologics, design, construction, and use of chromatography equipment; Class 042: Development of pharmaceutical preparations and medicines, namely, development services for others for the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development; consulting services for others concerning the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, design and construction of chromatography equipment, and design of biotech, biopharmaceutical and plasma manufacturing facilities, namely, scientific research and evaluation, and engineering; Design of biotech, biopharmaceutical and plasma manufacturing facilities relevant to the manufacturing of biolotics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate
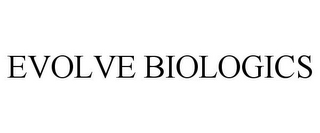
Owner:
Serial:87671420
Filed:Nov 3, 2017
Classes:5, 37, 40, +2
Registration:6297746
Registered:Mar 23, 2021
Goods & Services
Class 005: Biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for human clinical use, namely, plasma protein fractions, serum albumin, immunoglobulins, coagulation factors, antihemophilic factors, clotting factors, enzymes, coenzymes, coactivators, inhibitors, and structural proteins; resins, for medical use, namely, polysaccharide beads cross-linked to ligands for capturing proteins from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate using chromatographic methods; Class 037: Construction of biotech, biopharmaceutical and plasma manufacturing facilities relevant to the manufacturing of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate; consulting services concerning construction of biotech; Consulting services concerning building construction of biotech facilities; Class 040: Contract manufacturing services for others for biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, and clinical use; consulting services concerning manufacturing of biologics; Class 041: Educational services, namely, designing, arranging, and conducting training programs for research and development, manufacturing of biologics, design, construction, and use of chromatography equipment; Class 042: Development of pharmaceutical preparations and medicines, namely, development services for others for the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development; consulting services for others concerning the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, design and construction of chromatography equipment, and design of biotech, biopharmaceutical and plasma manufacturing facilities, namely, scientific research and evaluation, and engineering; Design of biotech, biopharmaceutical and plasma manufacturing facilities relevant to the manufacturing of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate

Owner:
Serial:86039785
Filed:Aug 16, 2013
Classes:1, 5, 9, +4
Registration:6172150
Registered:Oct 13, 2020
Goods & Services
Resins, namely, polysaccharide beads cross-linked to ligands for capturing proteins from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate ion exchange, mixed mode, and affinity resins | Biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for human clinical use, namely, plasma protein fractions, serum albumin, immunoglobulins, coagulation factors, antihemophilic factors, clotting factors, enzymes, coenzymes, coactivators, inhibitors, and structural proteins | Protein chromatography equipment for capturing biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate | Construction of biopharmaceutical and plasma manufacturing facilities relevant to the manufacturing of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate; consulting services in the field of construction of biopharmaceutical and plasma manufacturing services | Contract manufacturing services for others for biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, and clinical use; consulting services concerning manufacturing of biologics | Educational services, namely, designing, arranging, and conducting training programs for research and development, manufacturing of biologics, design, construction, and use of chromatography equipment | Development of pharmaceutical preparations and medicines, namely, development services for others for the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development; consulting services for others concerning the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, design of chromatography equipment, and design of biopharmaceutical and plasma manufacturing facilities, namely, scientific research and evaluation, and engineering

Owner:
Serial:86019797
Filed:Jul 25, 2013
Classes:1, 5, 9, +4
Registration:6172148
Registered:Oct 13, 2020
Goods & Services
Class 001: Resins, namely, polysaccharide beads cross-linked to ligands for capturing proteins from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate ion exchange, mixed mode, and affinity resins; Class 005: Biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for human clinical use, namely, plasma protein fractions, serum albumin, immunoglobulins, coagulation factors, antihemophilic factors, clotting factors, enzymes, coenzymes, coactivators, inhibitors, and structural proteins; Class 009: Protein chromatography equipment for capturing biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate; Class 037: Construction of biopharmaceutical and plasma manufacturing facilities relevant to the manufacturing of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate; consulting services in the field of construction of biopharmaceutical and plasma manufacturing services; Class 040: Contract manufacturing services for others for biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, and clinical use; consulting services concerning manufacturing of biologics; Class 041: Educational services, namely, designing, arranging, and conducting training programs for research and development, manufacturing of biologics, design, construction, and use of chromatography equipment; Class 042: Development of pharmaceutical preparations and medicines, namely, development services for others for the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development; consulting services for others concerning the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, design of chromatography equipment, and design of biopharmaceutical and plasma manufacturing facilities, namely, scientific research and evaluation, and engineering
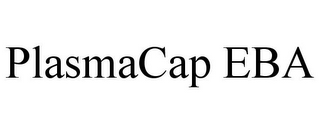
Owner:
Serial:86019802
Filed:Jul 25, 2013
Classes:1, 5, 9, +4
Registration:6172149
Registered:Oct 13, 2020
Goods & Services
Class 001: Resins, namely, polysaccharide beads cross-linked to ligands for capturing proteins from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate ion exchange, mixed mode, and affinity resins; Class 005: Biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for human clinical use, namely, plasma protein fractions, serum albumin, immunoglobulins, coagulation factors, antihemophilic factors, clotting factors, enzymes, coenzymes, coactivators, inhibitors, and structural proteins; Class 009: Protein chromatography equipment for capturing biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate; Class 037: Construction of biopharmaceutical and plasma manufacturing facilities relevant to the manufacturing of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate; consulting services in the field of construction of biopharmaceutical and plasma manufacturing services; Class 040: Contract manufacturing services for others for biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, and clinical use; consulting services concerning manufacturing of biologics; Class 041: Educational services, namely, designing, arranging, and conducting training programs for research and development, manufacturing of biologics, design, construction, and use of chromatography equipment; Class 042: Development of pharmaceutical preparations and medicines, namely, development services for others for the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development; consulting services for others concerning the capture of biologics from human blood, human blood plasma, fractionated human blood plasma, cryopaste, and cryoprecipitate for research and development, design of chromatography equipment, and design of biopharmaceutical and plasma manufacturing facilities, namely, scientific research and evaluation, and engineering