
FORMEDIX LTD.
4 Marks
Other
First Filed:Oct 3, 2011Latest Filed:Sep 3, 2020Address:The Connect Building, Glasgow, G22DH, SC
Portfolio Overview
Registered
2(50%)Dead
2(50%)Top Classes
Class 42
Software, IT Services & Scientific ResearchClass 9
Electronics, Software & Scientific EquipmentClass 35
Advertising, Business Services & RetailTTAB Proceedings
No litigation activity
Top Firms
Day Pitney LLP(Sep 2020)
2
Gesmer Updegrove LLP(Oct 2011)
1
STEIN IP LLC(Mar 2020)
1
Trademark Portfolio
4 results
(current & previous marks)
Refine by Mark/Description🔽
Filed
Registered
Last Activity
Filters
Ownership
4
0
Status
2
0
1
1
0
Mark Type
3
0
0
1
0
2
2
2
2

Goods & Services
Providing training services to enable pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to use clinical metadata management software platforms for managing clinical trial metadata and clinical trial data; providing training services to enable pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to collect the required clinical data and meet the regulations and standards required by international and government agencies for data collection, tabulation, analysis, and submission for clinical trials; providing training services in relation to the use of clinical metadata management computer software for use in relation to clinical trials; providing training services in relation to the use of clinical management computer software for use in relation to clinical trials; all of the aforesaid training services being used in relation to clinical trials only; On-line retail ordering services featuring clinical metadata management computer software for use in relation to clinical trials, clinical management computer software for use in relation to clinical trials, computer software for use in exclusively collecting, storing, controlling, managing and submitting of clinical metadata and clinical data in relation to clinical trials, database management computer software for use exclusively in relation to clinical trials, computer software to allow users to collect, store, control, manage and submit clinical metadata and clinical data in relation to clinical trials, computer databases for collecting, storing, controlling, managing and submitting clinical data in relation to clinical trials, computer software designed to ensure that clinical metadata and clinical data meets the regulations and standards required by international and government agencies in relation to clinical trials, computer software used by pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to help collect the required clinical data and meet the required standards for data collection, tabulation, analysis, and submission for clinical trials, all of the aforesaid computer software being used in relation to clinical trials only; Providing temporary use of on-line non-downloadable clinical metadata management computer software for use in relation to clinical trials; providing temporary use of on-line non-downloadable clinical management computer software for use in relation to clinical trials; providing temporary use of on-line non-downloadable computer software for use in exclusively collecting, storing, controlling, managing and submitting of clinical metadata and clinical data in relation to clinical trials; providing temporary use of on-line non-downloadable database management computer software for use exclusively in relation to clinical trials; providing temporary use of on-line non-downloadable computer software to allow users to collect, store, control, manage and submit clinical metadata and clinical data in relation to clinical trials; providing on-line computer databases for collecting, storing, controlling, managing and submitting clinical data in relation to clinical trials; providing temporary use of on-line non-downloadable computer software designed to ensure that clinical metadata and clinical data meets the regulations and standards required by international and government agencies in relation to clinical trials; providing temporary use of on-line non-downloadable computer software used by pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to help collect the required clinical data and meet the required standards for data collection, tabulation, analysis, and submission for clinical trials; design and development of computer software exclusively for managing clinical metadata and clinical data in clinical trials; designing, creating and maintaining web pages exclusively in connection with collecting, storing, controlling and managing clinical metadata and clinical data, to meet the regulations and standards required by international and government agencies in conducting clinical trials; all of the aforesaid software services being used in relation to clinical trials only; technical support services, namely, troubleshooting of computer software problems related to clinical metadata management software platforms for managing clinical trial metadata and clinical trial data; providing medical and scientific research information in the field of clinical trials to enable pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to collect the required clinical data and meet the regulations and standards required by international and government agencies for data collection, tabulation, analysis, and submission for clinical trials; technical support services, namely, troubleshooting of computer software problems related to clinical metadata management computer software for use in relation to clinical trials; technical support services, namely, troubleshooting of computer software problems related to clinical management computer software for use in relation to clinical trials; all of the aforesaid information and support services being used in relation to clinical trials only
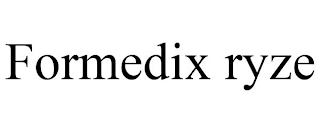
Owner:
Serial:90132747
Filed:Aug 24, 2020
Classes:35, 41, 42
Registration:6482181
Registered:Sep 14, 2021
Goods & Services
Providing training services to enable pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to use clinical metadata management software platforms for managing clinical trial metadata and clinical trial data; providing training services to enable pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to collect the required clinical data and meet the regulations and standards required by international and government agencies for data collection, tabulation, analysis, and submission for clinical trials; providing training services in relation to the use of clinical metadata management computer software for use in relation to clinical trials; providing training services in relation to the use of clinical management computer software for use in relation to clinical trials; all of the aforesaid training services being used in relation to clinical trials only; On-line retail ordering services featuring clinical metadata management computer software for use in relation to clinical trials, clinical management computer software for use in relation to clinical trials, computer software for use in exclusively collecting, storing, controlling, managing and submitting of clinical metadata and clinical data in relation to clinical trials, database management computer software for use exclusively in relation to clinical trials, computer software to allow users to collect, store, control, manage and submit clinical metadata and clinical data in relation to clinical trials, computer databases for collecting, storing, controlling, managing and submitting clinical data in relation to clinical trials, computer software designed to ensure that clinical metadata and clinical data meets the regulations and standards required by international and government agencies in relation to clinical trials, computer software used by pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to help collect the required clinical data and meet the required standards for data collection, tabulation, analysis, and submission for clinical trials, all of the aforesaid computer software being used in relation to clinical trials only; Providing temporary use of on-line non-downloadable clinical metadata management computer software for use in relation to clinical trials; providing temporary use of on-line non-downloadable clinical management computer software for use in relation to clinical trials; providing temporary use of on-line non-downloadable computer software for use in exclusively collecting, storing, controlling, managing and submitting of clinical metadata and clinical data in relation to clinical trials; providing temporary use of on-line non-downloadable database management computer software for use exclusively in relation to clinical trials; providing temporary use of on-line non-downloadable computer software to allow users to collect, store, control, manage and submit clinical metadata and clinical data in relation to clinical trials; providing on-line computer databases for collecting, storing, controlling, managing and submitting clinical data in relation to clinical trials; providing temporary use of on-line non-downloadable computer software designed to ensure that clinical metadata and clinical data meets the regulations and standards required by international and government agencies in relation to clinical trials; providing temporary use of on-line non-downloadable computer software used by pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to help collect the required clinical data and meet the required standards for data collection, tabulation, analysis, and submission for clinical trials; design and development of computer software exclusively for managing clinical metadata and clinical data in clinical trials; designing, creating and maintaining web pages exclusively in connection with collecting, storing, controlling and managing clinical metadata and clinical data, to meet the regulations and standards required by international and government agencies in conducting clinical trials; all of the aforesaid software services being used in relation to clinical trials only; technical support services, namely, troubleshooting of computer software problems related to clinical metadata management software platforms for managing clinical trial metadata and clinical trial data; providing medical and scientific research information in the field of clinical trials to enable pharmaceutical companies, biotechnology companies, medical device manufacturers, and contract research organizations to collect the required clinical data and meet the regulations and standards required by international and government agencies for data collection, tabulation, analysis, and submission for clinical trials; technical support services, namely, troubleshooting of computer software problems related to clinical metadata management computer software for use in relation to clinical trials; technical support services, namely, troubleshooting of computer software problems related to clinical management computer software for use in relation to clinical trials; all of the aforesaid information and support services being used in relation to clinical trials only

