
Quotient Sciences Ltd
6 Marks
Other
First Filed:Mar 3, 2016Latest Filed:Sep 17, 2024Address:Trent House Mere Way, Ruddington, Nottingham, NG116JS, GB
Portfolio Overview
Registered
5(83%)Pending
1(17%)Top Classes
Class 42
Software, IT Services & Scientific ResearchClass 39
Transportation and StorageClass 40
Material TreatmentTTAB Proceedings
No litigation activity
Top Firms
LAW OFFICE OF AMY COHEN HELL...(Mar 2021)
2
Sandberg Phoenix and von Gon...(Sep 2024)
2
The Belles Group, P.C.(Nov 2021)
1
Womble Bond Dickinson (US) L...(Mar 2016)
1
Trademark Portfolio
6 results
(current & previous marks)
Refine by Mark/Description🔽
Filed
Registered
Last Activity
Filters
Ownership
6
0
Status
5
1
0
0
0
Mark Type
4
1
0
1
0
3
1
4

Goods & Services
Custom manufacture of pharmaceuticals and biopharmaceuticals; custom manufacture of pharmaceuticals and biopharmaceuticals for clinical trial purposes; manufacturing services for clinical trials, namely manufacture of pharmaceuticals and biopharmaceuticals to order and/or specification of others; providing information about the results of clinical trials for pharmaceutical and biopharmaceutical products, namely providing technical information in the field of pharmaceutical and biopharmaceutical manufacturing and testing.; Contract packaging services in the nature of packaging and labelling of pharmaceutical and biopharmaceuticals for distribution on behalf of third parties; packaging and labelling of products for others, namely, contract packaging services in the nature of merchandise packaging for others; storage services, namely, warehousing and temperature controlled storage of goods for others; transportation logistics services, namely, arranging transportation of goods for others; packaging articles to the order and specification of others; packaging of pharmaceutical, biopharmaceutical and clinical supplies and materials, namely, contract packaging services in the nature of pharmaceutical, biopharmacutical and clinical supplies and materials packaging for others.; Clinical research in the field of pharmacology and medical research; clinical trials for others; conducting clinical trials for others; conducting clinical trials for others in the field of pharmaceutical and biopharmaceutical products; pharmaceutical and biopharmaceutical research services; pharmaceutical and biopharmaceutical testing services; research relating to science namely, scientific research, basic and clinical research in the fields of pharmacology, pharmaceuticals and biopharmaceuticals; analytical services, namely, evaluation and analysis of the efficacy of pharmaceutical and biopharmaceutical products and dosages in the nature of analysis and evaluation of pharmaceutical and biopharmaceutical products and dosages of others to determine conformity with certification standards and to assure compliance with industry standards; pharmaceutical and biopharmaceutical product evaluation; providing medical and scientific research information in the field of pharmaceuticals, biopharmaceuticals and clinical trials; biological research; research and development of vaccines and medicines; pharmaceutical and biopharmaceutical drug development services; pharmaceutical and biopharmaceutical products development; pharmaceutical and biopharmaceutical formulation design and development services, namely development of pharmaceutical and biopharmaceutical preparations and medicines; pre-formulation screening services, namely conducting early evaluations in the field of new pharmaceutical and testing, inspection and research of pharmaceuticals; conducting clinical trials for others for optimisation of pharmaceutical and biopharmaceutical formulations and dosage through clinical testing; development of pharmaceutical and biopharmaceutical preparations and medicines, research and development of vaccines and medicines, product research and development, product development for others, research relating to medicine, namely, medical research, basic and clinical research in the field of pharmacology; consultancy relating to pharmaceutical and biopharmaceutical testing and screening programs in particular testing, inspection and research of pharmaceuticals and biopharmaceuticals; consultancy in the field of pharmacology; clinical pharmacology, namely technical research, testing and consulting in the field of pharmaceutical and biopharmaceutical studies; laboratory research services relating to pharmaceuticals.

Goods & Services
Research services relating to the pharmaceutical, biotechnology and nutraceutical industry
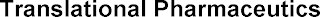
Goods & Services
Research services relating to the pharmaceutical industry

Goods & Services
Contract packaging services in the nature of packaging and labeling of pharmaceutical and biopharmaceuticals for distribution on behalf of third parties; packaging and labeling of products for others, namely, contract packaging services in the nature of merchandise packaging for others; storage services, namely, warehousing and temperature controlled storage of goods for others; transportation logistics services, namely, arranging transportation of goods for others; packaging articles to the order and specification of others; packaging of pharmaceutical, biopharmaceutical and clinical supplies and materials, namely, contract packaging services in the nature of pharmaceutical, biopharmaceutical and clinical supplies and materials packaging for others | Custom manufacture of pharmaceuticals and biopharmaceuticals; custom manufacture of pharmaceuticals and biopharmaceuticals for clinical trial purposes; manufacturing services for clinical trials, namely, manufacture of pharmaceuticals and biopharmaceuticals to order and/or specification of others; providing information about the results of clinical trials for pharmaceutical and biopharmaceutical products, namely, providing technical information in the field of pharmaceutical and biopharmaceutical manufacturing and testing | Clinical research in the field of pharmacology and medical research; clinical trials for others; conducting clinical trials for others; conducting clinical trials for others in the field of pharmaceutical and biopharmaceutical products; pharmaceutical and biopharmaceutical research services; pharmaceutical and biopharmaceutical testing services; research relating to science, namely, scientific research, basic and clinical research in the fields of pharmacology, pharmaceuticals and biopharmaceuticals; analytical services, namely, evaluation and analysis of the efficacy of pharmaceutical and biopharmaceutical products and dosages in the nature of analysis and evaluation of pharmaceutical and biopharmaceutical products and dosages to determine conformity with certification standards and to assure compliance with industry standards; pharmaceutical and biopharmaceutical product evaluation; providing medical and scientific research information in the fields of pharmaceuticals, biopharmaceuticals and clinical trials; biological research; research and development of vaccines and medicines; pharmaceutical and biopharmaceutical drug development services; pharmaceutical and biopharmaceutical products development; pharmaceutical and biopharmaceutical formulation design and development services, namely, development of pharmaceutical and biopharmaceutical preparations and medicines; pre-formulation screening services, namely, conducting early evaluations in the field of new pharmaceuticals and testing, inspection and research of pharmaceuticals; conducting clinical trials for others for optimisation of pharmaceutical and biopharmaceutical formulations and dosage through clinical testing; development of pharmaceutical and biopharmaceutical preparations and medicines, research and development of vaccines and medicines, product research and development, product development for others, research relating to medicine, namely, medical research, basic and clinical research in the field of pharmacology; consultancy relating to pharmaceutical and biopharmaceutical testing and screening programs in particular testing, inspection and research of pharmaceutical and biopharmaceuticals; consultancy in the field of pharmacology; clinical pharmacology, namely, technical research, testing and consulting in the field of pharmaceutical and biopharmaceutical studies; laboratory research services relating to pharmaceuticals

Goods & Services
Contract packaging services in the nature of packaging and labeling of pharmaceutical and biopharmaceuticals for distribution on behalf of third parties; packaging and labeling of products for others, namely, contract packaging services in the nature of merchandise packaging for others; storage services, namely, warehousing and temperature controlled storage of goods for others; transportation logistics services, namely, arranging transportation of goods for others; packaging articles to the order and specification of others; packaging of pharmaceutical, biopharmaceutical and clinical supplies and materials, namely, contract packaging services in the nature of pharmaceutical, biopharmaceutical and clinical supplies and materials packaging for others | Custom manufacture of pharmaceuticals and biopharmaceuticals; custom manufacture of pharmaceuticals and biopharmaceuticals for clinical trial purposes; manufacturing services for clinical trials, namely, manufacture of pharmaceuticals and biopharmaceuticals to order and/or specification of others; providing information about the results of clinical trials for pharmaceutical and biopharmaceutical products, namely, providing technical information in the field of pharmaceutical and biopharmaceutical manufacturing and testing | Clinical research in the field of pharmacology and medical research; clinical trials for others; conducting clinical trials for others; conducting clinical trials for others in the field of pharmaceutical and biopharmaceutical products; pharmaceutical and biopharmaceutical research services; pharmaceutical and biopharmaceutical testing services; research relating to science, namely, scientific research, basic and clinical research in the fields of pharmacology, pharmaceuticals and biopharmaceuticals; analytical services, namely, evaluation and analysis of the efficacy of pharmaceutical and biopharmaceutical products and dosages in the nature of analysis and evaluation of pharmaceutical and biopharmaceutical products and dosages of others to determine conformity with certification standards and to assure compliance with industry standards; pharmaceutical and biopharmaceutical product evaluation; providing medical and scientific research information in the field of pharmaceuticals, biopharmaceuticals and clinical trials; biological research; research and development of vaccines and medicines; pharmaceutical and biopharmaceutical drug development services; pharmaceutical and biopharmaceutical products development; pharmaceutical and biopharmaceutical formulation design and development services, namely, development of pharmaceutical and biopharmaceutical preparations and medicines; pre-formulation screening services, namely, conducting early evaluations in the field of new pharmaceutical and testing, inspection and research of pharmaceuticals; conducting clinical trials for others for optimisation of pharmaceutical and biopharmaceutical formulations and dosage through clinical testing; development of pharmaceutical and biopharmaceutical preparations and medicines, research and development of vaccines and medicines, product research and development, product development for others, research relating to medicine, namely, medical research, basic and clinical research in the field of pharmacology; consultancy relating to pharmaceutical and biopharmaceutical testing and screening programs in particular testing, inspection and research of pharmaceuticals and biopharmaceuticals; consultancy in the field of pharmacology; clinical pharmacology, namely, technical research, testing and consulting in the field of pharmaceutical and biopharmaceutical studies; laboratory research services relating to pharmaceuticals
