
SYNEOS HEALTH, LLC
17 Marks
LLC
First Filed:Dec 12, 2017Latest Filed:Sep 15, 2022Address:1030 Sync Street, Morrisville, NC 27560
Portfolio Overview
Registered
12(71%)Dead
5(29%)Top Classes
Class 35
Advertising, Business Services & RetailClass 42
Software, IT Services & Scientific ResearchClass 44
Medical, beauty and agricultural servicesTTAB Proceedings
Total Proceedings
5As Plaintiff
2(40%)As Defendant
3(60%)Top Firms
DLA Piper LLP (US)(Sep 2022)
17
Trademark Activity Timeline(2017 – 2024)
- Filings
- Registrations
Trademark Portfolio
17 results
(current & previous marks)
Refine by Mark/Description🔽
Filed
Registered
Last Activity
Filters
Ownership
17
0
Status
12
0
0
5
0
Mark Type
13
1
1
2
0
14
5
9
6
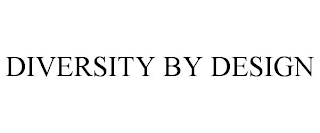
Goods & Services
Education in the field of pharmaceutical sales, namely, providing webinars, providing live workshops, providing on-line non-downloadable videos, providing on-line non-downloadable publications in the nature of articles and white papers, and providing podcasts; training services in the field of pharmaceutical sales; providing on-line non-downloadable newsletters and bulletins in the pharmaceutical, biotechnology and life sciences fields; educational services in the field of clinical trials, namely, providing on-line non-downloadable videos, webinars, live workshops, providing online non-downloadable publications in the nature of articles and white papers, and providing podcasts; Strategic business consulting, marketing and advertising, public relations communications strategy, information and data collection for business purposes, and business data analytics, namely, analyzing business data to understand and evaluate business performance, all of the aforementioned in the pharmaceutical, biotechnology and life sciences fields; sales promotion services; market analysis and research services; personnel recruitment and placement; recruitment services, namely, advertising clinical trials, recruiting patients, and placing patients for participation in clinical trials for testing of pharmaceuticals and devices; consulting in sales techniques and sales programs; sales volume tracking for others, all in the pharmaceutical, biotechnology and life sciences fields; business consulting in the field of clinical trials, namely, clinical data and regulatory submission management on behalf of medical, pharmaceutical and biotechnology companies to assist them with clinical research, clinical trials and applications for drug approval; assisting pharmaceutical and biotechnology companies with applications for new drugs with governmental regulatory bodies; promoting the exchange of health information to promote patient adherence; Providing medical information to physicians, other health care professionals and for medical patients; providing health information regarding the medications and health care products of others, namely, product discontinuations, new formulations, bottle changes, and seasonal information through e-mail alerts and push notifications; Medical, scientific and data analysis research services for others, namely, conducting and analyzing clinical trials in the pharmaceutical, biotechnology and life sciences fields; pharmaceutical drug development services, namely, developing clinical trial methodologies; consulting services in the area of pharmaceutical clinical testing; extended clinical research services for others, namely, computerized data management of clinical trials, biostatistical analysis, environmental consultancy, preclinical research and development evaluation, repairing flawed clinical trials and dossiers, strategic clinical research portfolio management, and biosurveillance monitoring; providing life science and scientific research information to physicians and other health care professionals, patients, and medical, pharmaceutical and biotechnology companies; providing regulatory compliant technical writing services in the field of medical research and clinical studies; consulting services in the field of contract clinical research and contract product development

Goods & Services
Strategic business consulting, marketing and advertising, public relations communications strategy, information and data collection for business purposes, and business data analytics, namely, analyzing business data to understand and evaluate business performance, all of the aforementioned in the pharmaceutical, biotechnology and life sciences fields; sales promotion services; market analysis and research services; personnel recruitment and placement; recruitment services, namely, advertising clinical trials, recruiting patients, and placing patients for participation in clinical trials for testing of pharmaceuticals and devices; consulting in sales techniques and sales programs; sales volume tracking for others, all in the pharmaceutical, biotechnology and life sciences fields; business consulting in the field of clinical trials, namely, clinical data and regulatory submission management on behalf of medical, pharmaceutical and biotechnology companies to assist them with clinical research, clinical trials and applications for drug approval; assisting pharmaceutical and biotechnology companies with applications for new drugs with governmental regulatory bodies; promoting the exchange of health information to promote patient adherence

Goods & Services
Class 035: Providing omnichannel marketing services and marketing strategy; consulting services for multichannel marketing services; business management consulting services, namely, providing analysis and analytics for marketing and advertising activities; providing business advice, consulting, and information in the field of marketing, namely, research, evaluations, and analytics related to digital advertising, audience participation, audience engagement and audience retention; all of the foregoing in the pharmaceutical, biotechnology and life sciences fields

Goods & Services
Class 042: Providing online non-downloadable software for storing, retrieving, accessing, displaying, managing, analyzing, and reviewing information and data for use in the fields of management of clinical trials and the development of pharmaceuticals

Owner:
Serial:90702195
Filed:May 11, 2021
Classes:35, 41, 42, +1
Registration:6686548
Registered:Mar 29, 2022
Goods & Services
Class 035: Strategic business consulting, marketing and advertising, public relations communications strategy, information and data collection for business purposes, and business data analytics, namely, analyzing business data to understand and evaluate business performance, all of the aforementioned in the pharmaceutical, biotechnology and life sciences fields; sales promotion services; market analysis and research services; personnel recruitment and placement; recruitment services, namely, advertising clinical trials, recruiting patients, and placing patients for participation in clinical trials for testing of pharmaceuticals and devices; consulting in sales techniques and sales programs; sales volume tracking for others, all in the pharmaceutical, biotechnology and life sciences fields; business consulting in the field of clinical trials, namely, clinical data and regulatory submission management on behalf of medical, pharmaceutical and biotechnology companies to assist them with clinical research, clinical trials and applications for drug approval; assisting pharmaceutical and biotechnology companies with applications for new drugs with governmental regulatory bodies; promoting the exchange of health information to promote patient adherence; Class 041: Education in the field of pharmaceutical sales, namely, providing webinars, providing live workshops, providing on-line non-downloadable videos, providing on-line non-downloadable publications in the nature of articles and white papers, and providing podcasts; training services in the field of pharmaceutical sales; providing on-line non-downloadable newsletters and bulletins in the pharmaceutical, biotechnology and life sciences fields; educational services in the field of clinical trials, namely, providing on-line non-downloadable videos, webinars, live workshops, providing online non-downloadable publications in the nature of articles and white papers, and providing podcasts; Class 042: Medical, scientific and data analysis research services for others, namely, conducting and analyzing clinical trials in the pharmaceutical, biotechnology and life sciences fields; pharmaceutical drug development services, namely, developing clinical trial methodologies; consulting services in the area of pharmaceutical clinical testing; extended clinical research services for others, namely, computerized data management of clinical trials, biostatistical analysis, environmental consultancy, preclinical research and development evaluation, repairing flawed clinical trials and dossiers, strategic clinical research portfolio management, and biosurveillance monitoring; providing life science and scientific research information to physicians and other health care professionals, patients, and medical, pharmaceutical and biotechnology companies; providing regulatory compliant technical writing services in the field of medical research and clinical studies; consulting services in the field of contract clinical research and contract product development; Internet development, design and consulting services for others, namely, creating websites featuring blogs about medical and pharmaceutical information related to chronic illnesses and disease, creating an online community for patients and caregivers to communicate about healthcare, and design and development of interactive software for use in the communication and dissemination of medical and pharmaceutical information; Class 044: Providing medical information to physicians, other health care professionals and for medical patients; providing health information regarding the medications and health care products of others, namely, product discontinuations, new formulations, bottle changes, and seasonal information through e-mail alerts and push notifications



Goods & Services
Business consulting services in the field of managed pharmaceutical and healthcare markets; advertising and brand development services for managed pharmaceutical and healthcare markets; business consulting services, namely, analytics services in the field of managed pharmaceutical and healthcare markets

Goods & Services
Strategic business consulting and data collection for business purposes, and business data analytics, namely, analyzing business data to understand and evaluate business performance, all of the aforementioned in the pharmaceutical, biotechnology and life sciences fields

Goods & Services
Business consulting and management in the field of clinical trials with regard to the burden experienced by patients of clinical trials; consulting services for others in the field of business project management of clinical trials | Providing medical and scientific research information in the field of clinical trials; consulting services for others in the field of design, planning and implementation project management of clinical trials

Goods & Services
business consulting services related to technology, design, development, clinical testing and product launch in the fields of pharmaceuticals, biotechnology and life sciences; business consulting services, namely, consulting services related to the use of data analytics and insights for strategy, marketing, sales, operation, and product design

Owner:
Serial:88428843
Filed:May 14, 2019
Classes:35, 41, 42, +1
Registration:6161365
Registered:Sep 29, 2020
Goods & Services
Strategic business consulting, information and data collection, namely, compiling and analyzing data and information for business purposes, and business data analytics, namely, analyzing business data to understand and evaluate business performance, all of the aforementioned in the pharmaceutical, biotechnology and life sciences fields; market analysis and research services; personnel recruitment and placement; recruitment advertising services, namely, advertising clinical trials for the purpose of recruiting patients, and placing patients for participation in clinical trials for testing of pharmaceuticals and devices; consulting in sales techniques and sales programs; sales volume tracking for others, all in the pharmaceutical, biotechnology and life sciences fields; business consulting in the field of clinical trials, namely, clinical data and regulatory submission management on behalf of medical, pharmaceutical and biotechnology companies to assist them with clinical research, clinical trials and applications for drug approval; regulatory submission management, namely, assisting pharmaceutical and biotechnology companies with preparing and filing applications for new drugs with governmental regulatory bodies; promoting the exchange of health information within the pharmaceutical, biotechnology and life sciences communities to promote patient adherence within the field of pharmaceutical, biotechnology and life sciences clinical trials; extended clinical research services for others, namely, computerized data management of clinical trials | Education services, namely, providing non-downloadable webinars, providing live workshops, providing seminars through on-line non-downloadable videos, providing on-line non-downloadable electronic publications in the nature of articles and white papers, and providing non-downloadable podcasts, all the foregoing in the field of pharmaceutical sales; training services in the field of pharmaceutical sales; providing on-line non-downloadable electronic newsletters and bulletins in the pharmaceutical, biotechnology and life sciences fields; educational services, namely, providing courses offered through on-line non-downloadable videos, non-downloadable webinars, live workshops, providing online non-downloadable electronic publications in the nature of articles and white papers, and providing non-downloadable podcasts, all the foregoing in the field of clinical trials | Medical and scientific data analysis research services for others, namely, conducting and analyzing clinical trials in the pharmaceutical, biotechnology and life sciences fields; pharmaceutical drug development services, namely, developing clinical trial methodologies; consulting services in the area of pharmaceutical product clinical testing; extended clinical research services for others for medical purposes, namely, biostatistical analysis for scientific research purposes, preclinical research in the field of pharmaceuticals, biotechnology, and medical sciences, development evaluation, namely, clinical feasibility assessment of protocols and patient recruitment strategies, repairing flawed clinical trials and dossiers, namely, medical and scientific research in the nature of conducting clinical trials for others, and strategic clinical research portfolio management, namely, consulting services for others in the project management of clinical research; extended clinical research services for others in the field of pharmaceuticals, biotechnology, and life sciences, namely, biostatistical analysis for scientific research purposes; providing life science and scientific research information to physicians and other health care professionals, patients, and medical, pharmaceutical and biotechnology companies; providing regulatory compliant technical writing services in the field of medical research and clinical studies; consulting services in the field of contract clinical research and contract product development | Providing medical information to physicians, other health care professionals and medical patients; providing health information regarding the medications and health care products of others through e-mail alerts and push notifications, namely, providing information concerning product discontinuations; providing health information concerning new formulations of medications and health care products of others through e-mail alerts and push notifications, namely, providing information in the field of pharmaceutical products development; providing health information concerning bottle changes of medications and health care products of others through e-mail alerts and push notifications, namely, providing information in the field of bottle product design, and seasonal information in the nature of reports on pharmaceutical products development

Owner:
Serial:88428831
Filed:May 14, 2019
Classes:35, 41, 42, +1
Registration:6093742
Registered:Jul 7, 2020
Goods & Services
Education services in the field of pharmaceutical sales, namely, providing non-downloadable webinars, providing live workshops, providing seminars through on-line non-downloadable videos, providing on-line non-downloadable electronic publications in the nature of articles and white papers, and providing non-downloadable podcasts, all the foregoing in the field of pharmaceutical sales; training services in the field of pharmaceutical sales; providing on-line non-downloadable electronic newsletters and bulletins in the pharmaceutical, biotechnology and life sciences fields; educational services, namely, providing courses offered through on-line non-downloadable videos, non-downloadable webinars, live workshops, providing online non-downloadable electronic publications in the nature of articles and white papers, and providing non-downloadable podcasts, all the foregoing in the field of clinical trials; Strategic business consulting, marketing and advertising, consultancy regarding public relations communications strategy, information and data collection, namely, compiling and analyzing data and information for business purposes, and business data analytics, namely, analyzing business data to understand and evaluate business performance, all of the aforementioned in the pharmaceutical, biotechnology and life sciences fields; sales promotion services; market analysis and research services; personnel recruitment and placement; recruitment advertising services, namely, advertising clinical trials for the purpose of recruiting patients, and placing patients for participation in clinical trials for testing of pharmaceuticals and devices; consulting in sales techniques and sales programs; sales volume tracking for others, all in the pharmaceutical, biotechnology and life sciences fields; business consulting in the field of clinical trials, namely, clinical data and regulatory submission management on behalf of medical, pharmaceutical and biotechnology companies to assist them with clinical research, clinical trials and applications for drug approval; regulatory submission management, namely, assisting pharmaceutical and biotechnology companies with preparing and filing applications for new drugs with governmental regulatory bodies; promoting the exchange of health information within the pharmaceutical, biotechnology and life sciences communities to promote patient adherence within the field of pharmaceutical, biotechnology and life sciences clinical trials; extended clinical research services for others, namely, computerized data management of clinical trials; Providing medical information to physicians, other health care professionals and medical patients; providing health information regarding the medications and health care products of others through e-mail alerts and push notifications, namely, providing information concerning product discontinuations; providing health information concerning new formulations of medications and health care products of others through e-mail alerts and push notifications, namely, providing information in the field of pharmaceutical products development; providing health information concerning bottle changes of medications and health care products of others through e-mail alerts and push notifications, namely, providing information in the field of bottle product design, and seasonal information in the nature of reports on pharmaceutical products development; "BIOPHARMACEUTICAL" AND "MODEL"; Medical and scientific data analysis research services for others, namely, conducting and analyzing clinical trials in the pharmaceutical, biotechnology and life sciences fields; pharmaceutical drug development services, namely, developing clinical trial methodologies; consulting services in the area of pharmaceutical product clinical testing; extended clinical research services for others for medical purposes, namely, biostatistical analysis for scientific research purposes, preclinical research in the field of pharmaceuticals, biotechnology, and medical sciences, development evaluation, namely, clinical feasibility assessment of protocols and patient recruitment strategies, repairing flawed clinical trials and dossiers, namely, medical and scientific research in the nature of conducting clinical trials for others, and strategic clinical research portfolio management, namely, consulting services for others in the project management of clinical research; extended clinical research services for others in the field of pharmaceuticals, biotechnology, and life sciences, namely, biostatistical analysis for scientific research purposes; providing life science and scientific research information to physicians and other health care professionals, patients, and medical, pharmaceutical and biotechnology companies; providing regulatory compliant technical writing services in the field of medical research and clinical studies; consulting services in the field of contract clinical research and contract product development; Internet development, design and consulting services for others, namely, creating websites featuring blogs about medical and pharmaceutical information related to chronic illnesses and disease, creating an online community for patients and caregivers to communicate about healthcare, and design and development of interactive computer software for use in the communication and dissemination of medical and pharmaceutical information

Goods & Services
Software as a Service (SaaS) services featuring software that allows for interaction with and the search and manipulation of a repository of data and information relating to drugs, biologics, medical devices, and public health related products and events; providing online non-downloadable computer software for searching, updating and extracting data in the field of drugs, biologics, medical devices, and public health related products and events; providing online non-downloadable computer software for the collection, compilation, storage, organization, search, use and manipulation of data relating to drugs, biologics, medical devices, and public health related products and events; providing temporary use of nondownloadable software featuring educational content, tools and materials, namely, white papers, clinical studies, diagrams, and other publications relating to drugs, biologics, medical devices, and public health related products and events | providing an online searchable database featuring data and information in the field of drugs, biologics, medical devices, and public health related products and events

Owner:
Serial:87717521
Filed:Dec 12, 2017
Classes:35, 41, 42, +1
Registration:5814321
Registered:Jul 23, 2019
Goods & Services
Education in the field of pharmaceutical sales, namely, providing webinars, providing live workshops, providing on-line non-downloadable videos, providing on-line non-downloadable publications in the nature of articles and white papers, and providing podcasts; training services in the field of pharmaceutical sales; providing on-line non-downloadable newsletters and bulletins in the pharmaceutical, biotechnology and life sciences fields; educational services in the field of clinical trials, namely, providing on-line non-downloadable videos, webinars, live workshops, providing online non-downloadable publications in the nature of articles and white papers, and providing podcasts; Strategic business consulting, marketing and advertising, public relations communications strategy, information and data collection for business purposes, and business data analytics, namely, analyzing business data to understand and evaluate business performance, all of the aforementioned in the pharmaceutical, biotechnology and life sciences fields; sales promotion services; market analysis and research services; personnel recruitment and placement; recruitment services, namely, advertising clinical trials, recruiting patients, and placing patients for participation in clinical trials for testing of pharmaceuticals and devices; consulting in sales techniques and sales programs; sales volume tracking for others, all in the pharmaceutical, biotechnology and life sciences fields; business consulting in the field of clinical trials, namely, clinical data and regulatory submission management on behalf of medical, pharmaceutical and biotechnology companies to assist them with clinical research, clinical trials and applications for drug approval; assisting pharmaceutical and biotechnology companies with applications for new drugs with governmental regulatory bodies; promoting the exchange of health information to promote patient adherence; Providing medical information to physicians, other health care professionals and for medical patients; providing health information regarding the medications and health care products of others, namely, product discontinuations, new formulations, bottle changes, and seasonal information through e-mail alerts and push notifications; "HEALTH"; Medical, scientific and data analysis research services for others, namely, conducting and analyzing clinical trials in the pharmaceutical, biotechnology and life sciences fields; pharmaceutical drug development services, namely, developing clinical trial methodologies; consulting services in the area of pharmaceutical clinical testing; extended clinical research services for others, namely, computerized data management of clinical trials, biostatistical analysis, environmental consultancy, preclinical research and development evaluation, repairing flawed clinical trials and dossiers, strategic clinical research portfolio management, and biosurveillance monitoring; providing life science and scientific research information to physicians and other health care professionals, patients, and medical, pharmaceutical and biotechnology companies; providing regulatory compliant technical writing services in the field of medical research and clinical studies; consulting services in the field of contract clinical research and contract product development; Internet development, design and consulting services for others, namely, creating websites featuring blogs about medical and pharmaceutical information related to chronic illnesses and disease, creating an online community for patients and caregivers to communicate about healthcare, and design and development of interactive software for use in the communication and dissemination of medical and pharmaceutical information

